Balanced Equation For Hcl And Na2co3
listenit
Apr 03, 2025 · 5 min read
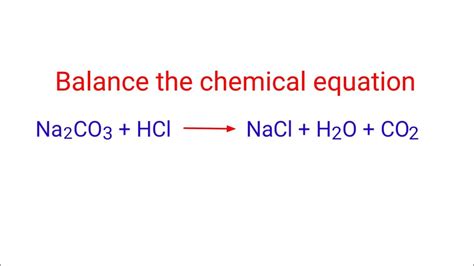
Table of Contents
The Balanced Equation for HCl and Na₂CO₃: A Comprehensive Exploration
The reaction between hydrochloric acid (HCl) and sodium carbonate (Na₂CO₃) is a classic example of an acid-base reaction, specifically an acid reacting with a carbonate. Understanding this reaction, its balanced equation, and its implications is crucial in various chemical contexts, from stoichiometry calculations to industrial applications. This article delves deep into the intricacies of this reaction, exploring its balanced equation, the stoichiometry involved, and the applications and implications of this chemical process.
Understanding the Reactants: HCl and Na₂CO₃
Before diving into the reaction itself, let's briefly examine the properties of the reactants: hydrochloric acid and sodium carbonate.
Hydrochloric Acid (HCl)
Hydrochloric acid is a strong, monoprotic acid. Strong means it completely dissociates in water, releasing hydrogen ions (H⁺) and chloride ions (Cl⁻). Monoprotic signifies it donates only one proton (H⁺) per molecule. This ability to readily donate protons is what makes it a strong acid and a key player in its reaction with sodium carbonate. HCl is commonly used in various industrial processes, laboratory experiments, and even as a component of stomach acid.
Sodium Carbonate (Na₂CO₃)
Sodium carbonate, also known as washing soda, is a salt of carbonic acid (H₂CO₃). It's a weak base, meaning it doesn't fully dissociate in water. However, it can react with acids, accepting protons. This acceptance of protons is the crucial element in its interaction with HCl. Sodium carbonate finds applications in water softening, detergent production, and various industrial processes.
The Reaction: HCl + Na₂CO₃
The reaction between HCl and Na₂CO₃ is an acid-base neutralization reaction. The hydrogen ions from the strong acid (HCl) react with the carbonate ion (CO₃²⁻) from the sodium carbonate, forming carbonic acid (H₂CO₃). However, carbonic acid is unstable and decomposes immediately into water (H₂O) and carbon dioxide (CO₂). This decomposition is a crucial aspect of the overall reaction.
Balancing the Chemical Equation
Balancing a chemical equation ensures that the number of atoms of each element is the same on both sides of the equation, adhering to the law of conservation of mass. The unbalanced equation for the reaction between HCl and Na₂CO₃ is:
HCl + Na₂CO₃ → NaCl + H₂O + CO₂
This equation is clearly unbalanced. To balance it, we need to adjust the coefficients (the numbers in front of the chemical formulas) to ensure the same number of each type of atom on both the reactant and product sides. The balanced equation is:
2HCl + Na₂CO₃ → 2NaCl + H₂O + CO₂
This balanced equation shows that two molecules of HCl react with one molecule of Na₂CO₃ to produce two molecules of NaCl, one molecule of H₂O, and one molecule of CO₂. The number of each type of atom is equal on both sides:
- Na: 2 on both sides
- Cl: 2 on both sides
- H: 2 on both sides
- C: 1 on both sides
- O: 3 on both sides
Stoichiometry and Calculations
The balanced equation provides the stoichiometric ratios between the reactants and products. This allows us to perform various calculations, such as determining the amount of product formed from a given amount of reactant or vice-versa. For example, if we know the number of moles of HCl reacting, we can use the stoichiometric ratio (2:1 HCl:Na₂CO₃) to determine the number of moles of Na₂CO₃ needed for complete reaction. Similarly, we can calculate the moles of NaCl, H₂O, and CO₂ produced.
Applications and Implications
The reaction between HCl and Na₂CO₃ has several practical applications:
Acid-Base Titrations
This reaction is frequently used in acid-base titrations to determine the concentration of either HCl or Na₂CO₃. By carefully measuring the volume of HCl required to neutralize a known amount of Na₂CO₃ (or vice versa), the concentration of the unknown solution can be calculated using the stoichiometry of the balanced equation.
Digestion and Dissolution
In analytical chemistry, the reaction can be utilized to dissolve carbonate-containing samples, aiding in further analysis. The reaction's efficiency in breaking down carbonate materials makes it a valuable tool for sample preparation in various analytical techniques.
Industrial Processes
In industrial settings, the reaction may occur as a side reaction or a planned process in applications involving both HCl and sodium carbonate. This can be involved in manufacturing processes or even in waste treatment, where the reaction's neutralization aspects play a role.
Understanding Chemical Reactions
Studying this specific reaction helps deepen the understanding of general acid-base chemistry, stoichiometry, and chemical equilibrium principles. It provides a clear example of a neutralization reaction and how to balance chemical equations properly.
Safety Precautions
When handling HCl and Na₂CO₃, it's crucial to follow standard laboratory safety procedures. HCl is corrosive and can cause burns. Na₂CO₃, while less hazardous, can still irritate skin and eyes. Always wear appropriate personal protective equipment (PPE), such as safety goggles, gloves, and a lab coat. Work in a well-ventilated area to avoid inhaling any fumes produced during the reaction.
Further Exploration
The reaction between HCl and Na₂CO₃ opens doors to further exploration of related chemical concepts. Investigating the kinetics of the reaction (how fast it proceeds), the influence of temperature and pressure on the reaction rate, and examining the equilibrium aspects can provide deeper insights into the chemical processes at play.
Conclusion
The reaction between hydrochloric acid and sodium carbonate is a fundamental acid-base reaction with significant implications in various fields. Understanding its balanced equation (2HCl + Na₂CO₃ → 2NaCl + H₂O + CO₂) and the stoichiometry involved is key to performing calculations and comprehending its applications in titrations, sample preparation, and industrial processes. By carefully examining the properties of the reactants and products and considering the safety precautions, one can gain a comprehensive understanding of this widely applicable chemical reaction. Remember to always prioritize safety when handling chemicals and to consult relevant safety data sheets (SDS) before conducting any experiments. The study of this seemingly simple reaction unlocks a deeper understanding of chemical principles and their practical applications in the real world.
Latest Posts
Latest Posts
-
What Is The Absolute Zero On The Celsius Scale
Apr 03, 2025
-
Meaning Of Trunk In Human Body
Apr 03, 2025
-
How Many Valence Electrons Are In Ar
Apr 03, 2025
-
Which Cell Organelle Controls The Activities Of The Entire Cell
Apr 03, 2025
-
Where Does Replication Occur In Eukaryotic Cells
Apr 03, 2025
Related Post
Thank you for visiting our website which covers about Balanced Equation For Hcl And Na2co3 . We hope the information provided has been useful to you. Feel free to contact us if you have any questions or need further assistance. See you next time and don't miss to bookmark.
