What Is The Absolute Zero On The Celsius Scale
listenit
Apr 03, 2025 · 5 min read
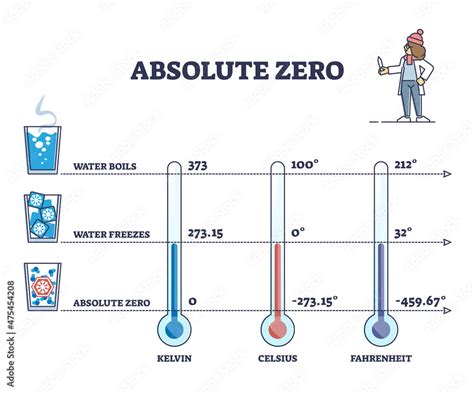
Table of Contents
What is Absolute Zero on the Celsius Scale?
Absolute zero is a crucial concept in physics and thermodynamics, representing the lowest possible temperature. While often discussed in relation to the Kelvin scale, understanding its equivalent on the Celsius scale is equally important. This article delves deep into the concept of absolute zero, explaining its significance, how it's determined, and its implications across various scientific fields. We'll explore its relationship with the Celsius scale, addressing common misconceptions and providing a comprehensive understanding of this fundamental physical limit.
Understanding Absolute Zero
Absolute zero is the point at which all molecular motion ceases. This doesn't mean that atoms and molecules stop moving entirely; quantum mechanical effects still prevent complete stillness. However, at absolute zero, the particles possess the minimum possible energy for their quantum state. This minimum energy is the zero point energy, and it's intrinsically linked to the uncertainty principle. In simpler terms, even at absolute zero, there's still some residual vibrational energy at a subatomic level.
The Significance of Absolute Zero
The significance of absolute zero stems from its implications for various scientific disciplines:
-
Thermodynamics: Absolute zero is the theoretical starting point for the thermodynamic temperature scale. It's the foundation upon which thermodynamic laws are built and is essential for understanding concepts like entropy and enthalpy.
-
Cryogenics: This branch of physics deals with extremely low temperatures. Cryogenics relies heavily on the understanding of absolute zero to develop technologies for achieving and utilizing these ultra-low temperatures. This includes applications in superconducting magnets, medical imaging (MRI), and materials science.
-
Quantum Mechanics: Absolute zero is crucial in understanding quantum phenomena. Many quantum effects, such as superfluidity and Bose-Einstein condensation, only manifest themselves at temperatures extremely close to absolute zero.
-
Cosmology: Understanding absolute zero is important in cosmological models. While the universe as a whole is not at absolute zero, the concepts of temperature and entropy are pivotal in explaining the evolution and ultimate fate of the universe.
Absolute Zero and the Kelvin Scale
The Kelvin scale, denoted by K, is the absolute thermodynamic temperature scale. Its zero point is defined as absolute zero, making it the most natural scale for representing temperature in many scientific contexts. This means that there are no negative temperatures on the Kelvin scale. An increase of 1 K is equivalent to an increase of 1 °C.
Calculating Absolute Zero on the Celsius Scale
The conversion between Kelvin and Celsius is straightforward:
K = °C + 273.15
Therefore, absolute zero (0 K) on the Celsius scale is:
°C = K - 273.15 = 0 - 273.15 = -273.15 °C
This means absolute zero is -273.15 degrees Celsius.
Why We Can't Reach Absolute Zero: The Third Law of Thermodynamics
The unattainability of absolute zero is a consequence of the Third Law of Thermodynamics, which states that the entropy of a perfect crystal approaches zero as the temperature approaches absolute zero. Entropy is a measure of disorder or randomness within a system. The Third Law implies that it is impossible to reach absolute zero in a finite number of steps. Even with advanced cooling techniques, we can only get increasingly closer, but never actually reach it.
Approaches to Achieving Extremely Low Temperatures
Despite the impossibility of reaching absolute zero, scientists have developed advanced techniques to achieve temperatures incredibly close to it:
-
Adiabatic Demagnetization: This technique involves repeatedly magnetizing and demagnetizing a paramagnetic salt, causing it to cool down.
-
Dilution Refrigeration: This method utilizes the properties of a mixture of helium-3 and helium-4 to achieve extremely low temperatures.
-
Laser Cooling: This technique employs lasers to slow down and cool atoms to temperatures approaching absolute zero.
Misconceptions about Absolute Zero
Several common misconceptions surround absolute zero:
-
Myth: Absolute zero means complete absence of energy. Reality: While the particles have minimal energy, quantum fluctuations and zero-point energy still exist.
-
Myth: Absolute zero represents a complete stop of all activity. Reality: Although macroscopic movement ceases, quantum effects prevent complete stillness at the subatomic level.
-
Myth: It's possible to reach absolute zero with advanced technology. Reality: The Third Law of Thermodynamics dictates the impossibility of reaching absolute zero in a finite number of steps.
Applications of Near-Absolute Zero Temperatures
The pursuit of near-absolute zero temperatures has led to numerous applications in various fields:
-
Superconductivity: Many materials exhibit superconductivity at extremely low temperatures, meaning they conduct electricity with zero resistance. This has implications for power transmission, magnetic levitation trains, and advanced medical imaging (MRI).
-
Bose-Einstein Condensation: At extremely low temperatures, certain atomic gases undergo Bose-Einstein condensation, a state of matter where a large fraction of atoms occupy the same quantum state. This state has applications in precision measurement and quantum computing.
-
Cryopreservation: Cryopreservation techniques, used to preserve biological materials at extremely low temperatures, rely on the principles of near-absolute zero temperatures.
Absolute Zero in Popular Culture
The concept of absolute zero has captured the imagination of writers, filmmakers, and science fiction authors. Often depicted as a place of extreme coldness and stillness, it serves as a symbol of ultimate limits and the mysteries of the universe. However, it’s important to remember that these depictions often creatively interpret the scientific reality for narrative purposes.
Conclusion
Absolute zero, at -273.15 °C or 0 K, represents a fundamental physical limit and a crucial concept in physics and thermodynamics. While it’s impossible to reach absolute zero, the pursuit of ever-lower temperatures has driven groundbreaking advancements in science and technology, leading to revolutionary applications in various fields. Understanding its implications, applications, and associated misconceptions is crucial for appreciating its profound significance in our understanding of the universe. From the behavior of matter at ultra-low temperatures to the laws governing the universe, absolute zero plays a vital role in our quest to understand the fundamental workings of the physical world. Its continued study promises further discoveries and innovations in years to come.
Latest Posts
Latest Posts
-
Write The Equation Of The Function Whose Graph Is Shown
Apr 04, 2025
-
Balanced Equation For Mg And Hcl
Apr 04, 2025
-
What Is The Molar Mass Of N
Apr 04, 2025
-
60 Of What Number Is 30
Apr 04, 2025
-
What Is 9 To The 0 Power
Apr 04, 2025
Related Post
Thank you for visiting our website which covers about What Is The Absolute Zero On The Celsius Scale . We hope the information provided has been useful to you. Feel free to contact us if you have any questions or need further assistance. See you next time and don't miss to bookmark.
