Where Is Most Of The Mass In An Atom Found
listenit
Apr 03, 2025 · 5 min read
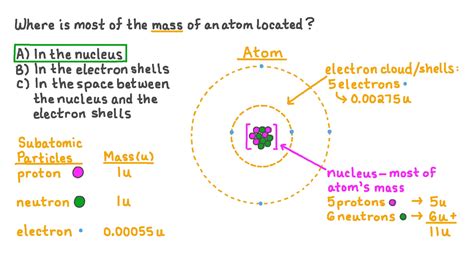
Table of Contents
Where is Most of the Mass in an Atom Found? A Deep Dive into Atomic Structure
Understanding where the mass of an atom resides is fundamental to grasping the nature of matter itself. While atoms are incredibly tiny, their internal structure is surprisingly complex, housing almost all their mass in a surprisingly small space. This article will delve into the intricacies of atomic structure, exploring the contributions of protons, neutrons, and electrons to an atom's overall mass and revealing where the bulk of this mass is concentrated.
The Atomic Model: A Brief Overview
Before we pinpoint the location of an atom's mass, let's briefly review the atomic model. Atoms, the basic building blocks of matter, consist of three primary subatomic particles:
- Protons: Positively charged particles residing in the atom's nucleus.
- Neutrons: Neutral particles (no charge) also located in the nucleus.
- Electrons: Negatively charged particles orbiting the nucleus in electron shells or orbitals.
The nucleus, the atom's central core, is incredibly dense, containing nearly all of the atom's mass. The electrons, while contributing to the atom's chemical properties, possess negligible mass compared to the protons and neutrons.
The Nucleus: The Mass Heavyweight Champion
The overwhelming majority of an atom's mass is concentrated within its nucleus. This tiny region, representing a minuscule fraction of the atom's overall volume, houses both protons and neutrons, collectively known as nucleons.
Protons: Positive Contributors to Mass
Protons carry a positive charge and contribute significantly to an atom's mass. Each proton possesses a mass of approximately 1.6726 x 10<sup>-27</sup> kilograms, which, while incredibly small, is substantial compared to the mass of an electron. The number of protons in an atom's nucleus determines its atomic number, which uniquely identifies the element. For example, hydrogen (H) has one proton, helium (He) has two, and so on.
Neutrons: Neutral Mass Players
Neutrons, as their name suggests, possess no electrical charge. Their mass is very slightly larger than that of a proton, approximately 1.6749 x 10<sup>-27</sup> kilograms. While they don't contribute to the atom's overall charge, they play a crucial role in stabilizing the nucleus and influencing an atom's isotopic properties. Isotopes are atoms of the same element with the same number of protons but a different number of neutrons.
Calculating Atomic Mass: The Role of Protons and Neutrons
The atomic mass of an atom is essentially the sum of the masses of its protons and neutrons. Electrons, being so much lighter, contribute almost negligibly to the total mass. This is why the atomic mass number, often rounded to the nearest whole number, is approximately equal to the sum of protons and neutrons. It's important to note that the actual atomic mass can vary slightly due to the binding energy of the nucleus (discussed later).
Electrons: Negligible Mass, Significant Influence
In contrast to the nucleus, electrons are incredibly lightweight. Each electron possesses a mass of approximately 9.1094 x 10<sup>-31</sup> kilograms, which is about 1836 times smaller than the mass of a proton. While their individual masses are insignificant compared to protons and neutrons, electrons play a critical role in determining an atom's chemical behavior and how it interacts with other atoms. They occupy specific energy levels or orbitals surrounding the nucleus, forming electron shells. The arrangement of electrons in these shells determines an element's chemical reactivity and its position in the periodic table.
Binding Energy: A Subtle Mass Reduction
The mass of a nucleus is slightly less than the sum of the masses of its individual protons and neutrons. This difference is due to the binding energy that holds the nucleons together. According to Einstein's famous equation, E=mc², energy and mass are equivalent and interchangeable. The energy required to overcome the strong nuclear force holding the nucleus together is converted into a small amount of mass. This mass defect, as it's known, represents the binding energy and contributes to the slight discrepancy between the expected and actual atomic mass. The higher the binding energy, the more stable the nucleus. This concept is crucial in understanding nuclear reactions, such as fission and fusion.
Comparing Mass Contributions: A Quantitative Perspective
To further illustrate the dominance of the nucleus in terms of mass, let's consider a specific example: the carbon-12 atom. Carbon-12 has six protons and six neutrons in its nucleus, along with six electrons orbiting the nucleus.
- Mass of protons (6 protons x 1.6726 x 10<sup>-27</sup> kg/proton): approximately 1.0036 x 10<sup>-26</sup> kg
- Mass of neutrons (6 neutrons x 1.6749 x 10<sup>-27</sup> kg/neutron): approximately 1.0049 x 10<sup>-26</sup> kg
- Total mass of protons and neutrons: approximately 2.0085 x 10<sup>-26</sup> kg
- Mass of electrons (6 electrons x 9.1094 x 10<sup>-31</sup> kg/electron): approximately 5.4656 x 10<sup>-30</sup> kg
As you can see, the mass of the electrons is several orders of magnitude smaller than the combined mass of protons and neutrons. The contribution of electrons to the overall mass is practically negligible. Therefore, the vast majority (over 99.9%) of the mass of a carbon-12 atom resides within its nucleus. This pattern holds true for almost all atoms.
Beyond Atomic Mass: Isotopes and Average Atomic Mass
While the atomic mass number provides a good approximation of an atom's mass, it's essential to consider the existence of isotopes. Many elements occur naturally as a mixture of isotopes, each possessing a different number of neutrons. The average atomic mass reported in the periodic table reflects the weighted average of the masses of all naturally occurring isotopes of an element, taking into account their relative abundances.
Practical Applications and Conclusion
Understanding where the majority of an atom's mass is located has far-reaching implications across numerous scientific fields. It's crucial for advancements in nuclear physics, nuclear medicine, materials science, and many other disciplines. Knowledge of atomic structure is essential for interpreting nuclear reactions, designing new materials with specific properties, and developing advanced technologies.
In conclusion, the vast majority of an atom's mass is concentrated in its nucleus, specifically within the protons and neutrons. The contribution of electrons to the overall mass is negligible. This fundamental understanding of atomic structure underpins our comprehension of the physical world and its various phenomena. The study of atomic mass and its distribution remains a cornerstone of scientific progress and technological innovation.
Latest Posts
Latest Posts
-
How Does An Igneous Rock Turn Into A Metamorphic Rock
Apr 04, 2025
-
What Is The Lcm Of 6 And 5
Apr 04, 2025
-
Which One Increases Number Of Collisions Between The Reactants
Apr 04, 2025
-
How Many Feet In 1 2 Miles
Apr 04, 2025
-
What Is 3 4 Of A Pound
Apr 04, 2025
Related Post
Thank you for visiting our website which covers about Where Is Most Of The Mass In An Atom Found . We hope the information provided has been useful to you. Feel free to contact us if you have any questions or need further assistance. See you next time and don't miss to bookmark.
