What Is The Horizontal Row In The Periodic Table
listenit
Apr 04, 2025 · 7 min read
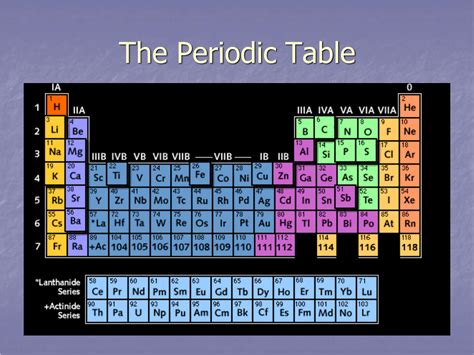
Table of Contents
What is a Horizontal Row in the Periodic Table? Understanding Periods and Their Significance
The periodic table, a cornerstone of chemistry, organizes elements based on their atomic structure and recurring chemical properties. While many are familiar with the table's overall structure, understanding the specific meaning and implications of its horizontal rows, known as periods, is crucial for grasping fundamental chemical concepts. This comprehensive guide delves into the intricacies of periods in the periodic table, exploring their significance in predicting element properties and behavior.
Understanding Periods: A Horizontal Journey Through Atomic Structure
A period in the periodic table represents a horizontal row of elements. Each period signifies a principal energy level or shell within an atom's electron configuration. The number of the period corresponds directly to the highest principal quantum number (n) of the electrons in an atom's ground state. For example, elements in Period 1 have electrons only in the n=1 shell, while elements in Period 2 have electrons in both the n=1 and n=2 shells.
This seemingly simple observation holds profound implications for understanding the properties of the elements. As we move across a period, the number of protons and electrons increases, leading to systematic changes in atomic size, ionization energy, electronegativity, and other crucial properties. These trends are predictable and help chemists understand how elements will interact with one another.
The Significance of Electron Shells
The organization of elements by period highlights the role of electron shells in determining chemical behavior. Electrons occupy specific energy levels or shells, and the outermost shell, known as the valence shell, plays a dominant role in chemical bonding. Elements within the same period have their valence electrons in the same principal energy level, although the number of valence electrons varies across the period. This shared valence shell is why elements within a period exhibit trends in their properties, despite differences in their number of protons and electrons.
Electron configuration, which describes the arrangement of electrons in an atom's shells and subshells, directly dictates the chemical behavior of an element. Elements in the same period may have different electron configurations in their subshells (s, p, d, f), but they all share a common highest principal quantum number. This commonality is the foundation of the periodic trends observed within a period.
Periodic Trends Within a Period: A Systematic Change
As we progress across a period from left to right, several key properties exhibit predictable trends. Understanding these trends provides valuable insight into element reactivity and behavior.
1. Atomic Radius: A Decrease Across the Period
Atomic radius, the distance from the nucleus to the outermost electrons, generally decreases as we move from left to right across a period. This is because the number of protons in the nucleus increases, leading to a stronger positive charge attracting the electrons more effectively. This stronger pull pulls the electrons closer to the nucleus, resulting in a smaller atomic radius. The addition of electrons to the same principal energy level does not significantly shield the outer electrons from the increased nuclear charge.
The decrease in atomic radius across a period is a significant factor affecting various properties, influencing the ease with which an atom can lose or gain electrons.
2. Ionization Energy: An Increase Across the Period
Ionization energy is the energy required to remove an electron from a neutral gaseous atom. It generally increases across a period from left to right. As the effective nuclear charge (the net positive charge experienced by valence electrons) increases across a period, it becomes more difficult to remove an electron, thereby increasing the ionization energy. The stronger attraction between the nucleus and the electrons necessitates greater energy input for electron removal.
This increasing trend in ionization energy reflects the increasing stability of the atoms as they approach a full outer electron shell (octet rule).
3. Electronegativity: A Rise Across the Period
Electronegativity is a measure of an atom's ability to attract electrons in a chemical bond. Similar to ionization energy, electronegativity generally increases across a period from left to right. The increasing effective nuclear charge leads to a stronger attraction for bonding electrons, hence the increase in electronegativity. Highly electronegative elements tend to attract electrons strongly in chemical bonds, forming polar or ionic compounds.
Electronegativity is crucial in understanding the nature of chemical bonds, determining whether a bond is covalent, polar covalent, or ionic.
4. Metallic Character: A Gradual Transition
Metallic character, referring to the properties typically associated with metals such as conductivity, malleability, and ductility, generally decreases across a period. Elements on the left side of a period are typically metals, while elements on the right are typically non-metals. This trend is linked to the ease with which atoms lose electrons. Metals readily lose electrons, while non-metals tend to gain electrons. The decrease in metallic character reflects the increasing tendency of atoms to gain electrons rather than lose them as we move across the period.
The Periods and the Block Structure: A Deeper Dive
The periodic table is further divided into blocks based on the subshells being filled. The relationships between periods and blocks provide a more nuanced understanding of electron configurations and chemical properties.
-
s-block: This block consists of the first two groups (alkali metals and alkaline earth metals) in each period. The valence electrons in these elements occupy the s subshell. Elements in the s-block are typically highly reactive metals.
-
p-block: This block consists of groups 13-18 and encompasses a wide range of elements, including some metals, metalloids, and non-metals. The valence electrons occupy the p subshell. The p-block elements display a greater diversity in properties compared to the s-block.
-
d-block: The d-block elements (transition metals) are found in the middle of the periodic table, occupying periods 4-7. They are characterized by the filling of the d subshells. Transition metals are known for their variable oxidation states and the formation of colorful complexes.
-
f-block: The f-block elements (lanthanides and actinides) are placed separately at the bottom of the periodic table. They are characterized by the filling of the f subshells. These elements have similar chemical properties within their respective series.
Period Length and Subshell Filling
The length of each period is determined by the number of electrons that can be accommodated in the subshells that are being filled in that period.
-
Periods 1 and 2: Short periods with lengths of 2 and 8, respectively, reflecting the filling of the 1s and 2s, and 2p subshells.
-
Periods 3 and 4: Periods 3 and 4 contain 8 and 18 elements, indicating the filling of the 3s and 3p, and 4s and 3d subshells respectively.
-
Periods 5 and 6: Periods 5 and 6 contain 18 and 32 elements, due to the filling of the 5s, 4d, and 5p, and 6s, 4f, 5d, and 6p subshells respectively.
-
Period 7: This period is incomplete but expected to eventually contain 32 elements, reflecting the filling of the 7s, 5f, 6d, and 7p subshells.
Predicting Properties Using Periodicity
The periodic table's organization allows us to predict the properties of elements based on their position within the periods. This is a powerful tool for chemists as it minimizes the need for individual characterization of each element. Knowing the period number immediately provides information about the valence shell and offers insight into potential reactivity and bonding behaviors. Combining the periodic trend within a period and the group trend (vertical columns) allows for highly accurate predictions of element properties.
Conclusion: The Unifying Principle of Periods
In conclusion, understanding periods in the periodic table is fundamental to grasping the organization and properties of elements. The horizontal rows reveal the systematic changes in atomic structure and properties as we progress across the table. The connection between periods, electron shells, and subshell filling provides a framework for predicting chemical behavior and interpreting chemical trends. This knowledge is not just an academic exercise; it forms the bedrock of countless applications in chemistry, materials science, and related fields. The consistent trends within each period are a testament to the remarkable organization and predictive power of the periodic table, a tool that continues to shape our understanding of the material world.
Latest Posts
Latest Posts
-
What Is The Si Unit Of Volume
Apr 05, 2025
-
What Organelle Is Dna Found In
Apr 05, 2025
-
Homologous Chromosomes Separate During Which Phase Of Meiosis
Apr 05, 2025
-
How Does An Outlier Affect The Mean
Apr 05, 2025
-
Why Do Elements In The Same Family Have Similar Properties
Apr 05, 2025
Related Post
Thank you for visiting our website which covers about What Is The Horizontal Row In The Periodic Table . We hope the information provided has been useful to you. Feel free to contact us if you have any questions or need further assistance. See you next time and don't miss to bookmark.
