What Is The Electron Configuration Of Ga
listenit
Apr 05, 2025 · 6 min read
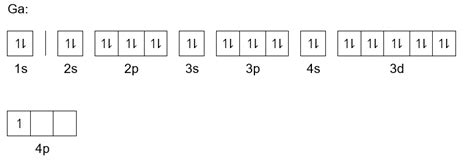
Table of Contents
What is the Electron Configuration of Ga? A Deep Dive into Gallium's Atomic Structure
Gallium (Ga), a fascinating element residing in Group 13 of the periodic table, boasts a unique electron configuration that dictates its chemical properties and behavior. Understanding this configuration is crucial to comprehending its role in various applications, from semiconductors to LEDs. This article delves deep into the electron configuration of gallium, explaining its derivation, implications, and relevance in different contexts.
Understanding Electron Configuration
Before we explore gallium's specific configuration, let's establish a foundational understanding of what electron configuration represents. An electron configuration describes how electrons are distributed among the various energy levels (shells) and sublevels (orbitals) within an atom. This distribution is governed by the Aufbau principle, Hund's rule, and the Pauli exclusion principle.
-
The Aufbau Principle: Electrons fill the lowest energy levels first. Think of it like filling a bucket from the bottom up – you wouldn't start filling the top until the bottom is full.
-
Hund's Rule: Within a sublevel (like a p or d orbital), electrons will individually occupy each orbital before pairing up. They prefer to have their own "space" before sharing.
-
Pauli Exclusion Principle: No two electrons in an atom can have the same set of four quantum numbers (n, l, ml, and ms). This means each orbital can hold a maximum of two electrons, with opposite spins.
These principles work together to predict the most stable and likely arrangement of electrons in an atom.
Deriving the Electron Configuration of Gallium (Ga)
Gallium has an atomic number of 31, meaning it has 31 protons and 31 electrons in its neutral state. To determine its electron configuration, we follow the Aufbau principle and fill the orbitals in order of increasing energy:
1s<sup>2</sup> 2s<sup>2</sup> 2p<sup>6</sup> 3s<sup>2</sup> 3p<sup>6</sup> 4s<sup>2</sup> 3d<sup>10</sup> 4p<sup>1</sup>
Let's break this down step-by-step:
-
1s<sup>2</sup>: The first shell (n=1) contains only one sublevel, the 's' orbital, which can hold a maximum of two electrons. Gallium's two lowest-energy electrons fill this orbital.
-
2s<sup>2</sup> 2p<sup>6</sup>: The second shell (n=2) contains an 's' orbital (holding two electrons) and three 'p' orbitals (holding a total of six electrons). These eight electrons fill the second shell completely.
-
3s<sup>2</sup> 3p<sup>6</sup>: The third shell (n=3) similarly contains an 's' orbital and three 'p' orbitals, accommodating another eight electrons.
-
4s<sup>2</sup>: The fourth shell begins with the 's' orbital, which holds two electrons. Note that the 4s orbital is lower in energy than the 3d orbital, hence filling it first.
-
3d<sup>10</sup>: After the 4s orbital is filled, the 3d orbitals, belonging to the third shell, are filled with ten electrons.
-
4p<sup>1</sup>: Finally, the remaining electron occupies one of the 4p orbitals.
Therefore, the complete electron configuration of gallium is 1s<sup>2</sup> 2s<sup>2</sup> 2p<sup>6</sup> 3s<sup>2</sup> 3p<sup>6</sup> 4s<sup>2</sup> 3d<sup>10</sup> 4p<sup>1</sup>.
Simplified Notation: Noble Gas Configuration
Writing out the full electron configuration can be lengthy. A more concise way to represent it is using the noble gas configuration. We find the noble gas that precedes gallium in the periodic table, which is argon (Ar). Argon has the electron configuration 1s<sup>2</sup> 2s<sup>2</sup> 2p<sup>6</sup> 3s<sup>2</sup> 3p<sup>6</sup>. We can replace this part of gallium's configuration with [Ar] to simplify:
[Ar] 4s<sup>2</sup> 3d<sup>10</sup> 4p<sup>1</sup>
This notation is much more compact and still conveys all the necessary information about the electron arrangement.
Implications of Gallium's Electron Configuration
Gallium's electron configuration is directly responsible for its chemical and physical properties. The presence of one electron in the 4p orbital is particularly significant:
-
Valence Electron: This single 4p electron is the valence electron, meaning it's the electron most likely to participate in chemical bonding. This explains gallium's +3 oxidation state, where it readily loses three electrons (the two 4s and one 4p electron) to form stable ions.
-
Metallic Character: The relatively loosely held valence electron contributes to gallium's metallic character. It is a soft, silvery metal with a low melting point. The delocalized electrons allow for good electrical and thermal conductivity.
-
Semiconductor Properties: The energy gap between the valence band (containing the valence electrons) and the conduction band (where electrons move freely to conduct electricity) in gallium is relatively small. This property makes gallium and its compounds, particularly gallium arsenide (GaAs), valuable semiconductors in electronic devices. The precise energy gap can be tuned through doping, further enhancing its semiconductor capabilities.
-
Formation of Compounds: The single valence electron allows gallium to readily form compounds with various elements, exhibiting diverse bonding characteristics (covalent, metallic, etc.) depending on the element it interacts with. Gallium's compounds find applications in diverse areas, including electronics, medicine, and materials science.
Gallium in Technology: A Look at Applications
Gallium's unique electron configuration translates into a range of crucial technological applications. Here are some examples:
-
Semiconductors: Gallium arsenide (GaAs) and other gallium-containing compounds are extensively used in high-speed transistors, integrated circuits, and solar cells. Their higher electron mobility compared to silicon makes them ideal for applications requiring faster switching speeds and higher frequencies.
-
LEDs and Lasers: Gallium nitride (GaN) is a key material in the production of high-efficiency light-emitting diodes (LEDs) and lasers. The specific wavelengths of light emitted can be controlled by adjusting the composition, allowing for the creation of various colors. GaN-based LEDs are increasingly replacing incandescent and fluorescent lighting due to their energy efficiency and longer lifespan.
-
Medical Applications: Gallium-67, a radioactive isotope of gallium, is used in nuclear medicine for imaging studies, particularly for detecting infections and tumors. Its tendency to accumulate in areas of inflammation makes it a useful diagnostic tool.
-
Other Applications: Gallium finds niche applications in various other fields, including aerospace, where its low melting point makes it suitable for certain types of alloys; and in the manufacturing of high-temperature thermometers.
Conclusion
The electron configuration of gallium ([Ar] 4s<sup>2</sup> 3d<sup>10</sup> 4p<sup>1</sup>) is not just a theoretical concept but a fundamental determinant of its properties and applications. Understanding this configuration allows us to appreciate the unique behavior of this element and its contribution to various technologies that shape modern life. From the high-speed electronics in our smartphones to the energy-efficient LEDs illuminating our homes, gallium's electron configuration plays a crucial role in driving technological advancements. Further research into the intricacies of gallium's electronic structure is likely to uncover new applications and enhance existing ones, highlighting the enduring importance of this seemingly simple element.
Latest Posts
Latest Posts
-
How Many Pi Bonds Are In A Triple Bond
Apr 05, 2025
-
How To Find Area Of Parallelogram With Vectors
Apr 05, 2025
-
Why Do Contour Lines Never Cross
Apr 05, 2025
-
Is Boron A Gas Solid Or Liquid
Apr 05, 2025
-
How Do You Factor 2x 2 7x 3
Apr 05, 2025
Related Post
Thank you for visiting our website which covers about What Is The Electron Configuration Of Ga . We hope the information provided has been useful to you. Feel free to contact us if you have any questions or need further assistance. See you next time and don't miss to bookmark.
