What Is The Correct Formula For Aluminum Oxide
listenit
Apr 03, 2025 · 5 min read
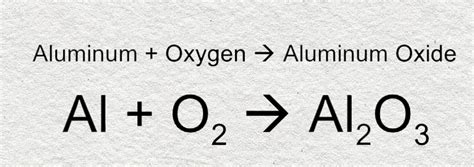
Table of Contents
What is the Correct Formula for Aluminum Oxide? A Deep Dive into Chemistry and Nomenclature
Aluminum oxide, a ubiquitous compound found in nature and extensively used in various industrial applications, holds a seemingly simple yet fascinating chemical story. While the formula Al₂O₃ is widely accepted and used, understanding its nuances requires delving into the intricacies of chemical bonding, crystal structures, and the complexities of chemical nomenclature. This article will explore the correct formula, its derivation, various forms, and its significance in diverse fields.
Understanding the Basics: Aluminum and Oxygen
Before diving into the formula, let's refresh our understanding of the constituent elements: aluminum (Al) and oxygen (O).
-
Aluminum (Al): Aluminum is a silvery-white, lightweight metal belonging to Group 13 (formerly IIIA) of the periodic table. Its atomic number is 13, meaning it possesses 13 protons and, in its neutral state, 13 electrons. In chemical reactions, aluminum readily loses three electrons to achieve a stable octet configuration, forming a +3 cation (Al³⁺). This tendency to lose three electrons is crucial in determining its bonding behavior with other elements.
-
Oxygen (O): Oxygen, a highly reactive non-metal, resides in Group 16 (formerly VIA) of the periodic table. Its atomic number is 8, indicating eight protons and eight electrons. To achieve a stable electron configuration, oxygen readily gains two electrons, forming a -2 anion (O²⁻). This strong electronegativity makes oxygen a highly effective oxidizing agent.
Deriving the Formula for Aluminum Oxide: Ionic Bonding
The formation of aluminum oxide (Al₂O₃) is a classic example of ionic bonding. The highly electropositive aluminum atom readily donates its three valence electrons to the highly electronegative oxygen atom. Each oxygen atom needs two electrons to complete its octet. Therefore, to balance the charges and achieve electrical neutrality, we need two aluminum atoms (each donating three electrons, for a total of six) and three oxygen atoms (each accepting two electrons, also for a total of six).
This charge balance can be represented mathematically:
2Al³⁺ + 3O²⁻ → Al₂O₃
This equation demonstrates that two aluminum ions (each with a +3 charge) combine with three oxide ions (each with a -2 charge) to form one formula unit of aluminum oxide, resulting in a neutral compound. This is why Al₂O₃ is the correct and most commonly used chemical formula for aluminum oxide.
Beyond the Simple Formula: Polymorphs and Crystal Structures
While Al₂O₃ represents the stoichiometric composition, aluminum oxide exists in various crystalline forms, known as polymorphs. These polymorphs exhibit different physical and chemical properties despite having the same chemical formula. The most common polymorphs include:
-
α-Alumina (Corundum): This is the most stable and thermodynamically favored form of aluminum oxide at standard temperature and pressure. It has a hexagonal close-packed structure, exhibiting high hardness, high melting point, and excellent chemical inertness. Corundum is the primary component of rubies and sapphires, the color being attributed to trace impurities like chromium (rubies) and titanium/iron (sapphires).
-
γ-Alumina (Gamma Alumina): This is a metastable form, meaning it's less stable than α-alumina and tends to convert to α-alumina upon heating. γ-alumina has a defective spinel structure and a much larger surface area compared to α-alumina. This high surface area makes it highly porous and suitable for use as a catalyst support, adsorbent, and desiccant.
-
Other polymorphs: Several other less common polymorphs exist, including θ-alumina, δ-alumina, and χ-alumina. Each polymorph exhibits a unique crystal structure and properties. The transition between these polymorphs is often temperature-dependent.
Applications of Aluminum Oxide: A Diverse Range
The diverse properties of aluminum oxide and its various polymorphs contribute to its widespread use across numerous industries:
-
Abrasives: α-alumina's high hardness makes it an excellent abrasive material in sandpaper, grinding wheels, and polishing compounds.
-
Refractories: Its high melting point and chemical resistance make aluminum oxide a critical component in refractory bricks used in high-temperature furnaces and kilns.
-
Ceramics: Aluminum oxide is a vital component in advanced ceramics due to its strength, hardness, and biocompatibility. It's used in dental implants, orthopedic components, and high-performance engineering ceramics.
-
Catalysis: γ-alumina's high surface area and porosity make it an ideal catalyst support in various chemical processes.
-
Electronics: Aluminum oxide is used as an insulator and dielectric material in electronic components.
-
Cosmetics: Aluminum oxide is a common ingredient in cosmetics and personal care products due to its mild abrasive properties and its ability to absorb moisture.
Nomenclature and Related Compounds
The name "aluminum oxide" is straightforward and reflects the composition of the compound. However, other names like alumina are also commonly used. These variations arise from historical usage and different contexts.
It's important to distinguish aluminum oxide from related compounds like aluminum hydroxide (Al(OH)₃) and aluminum oxyhydroxides, which have different compositions and properties. These compounds often arise through hydrolysis reactions involving aluminum oxide or aluminum salts.
Conclusion: The Significance of Al₂O₃
The formula Al₂O₃ accurately represents the stoichiometric ratio of aluminum and oxygen in aluminum oxide. However, it's crucial to acknowledge the existence of different polymorphs and the complexity of the crystal structures. The properties and applications of aluminum oxide are significantly influenced by its crystalline form and the presence of impurities. Understanding the chemical formula, crystal structures, and diverse applications of aluminum oxide is crucial across various scientific and engineering disciplines, solidifying its position as a vital material in modern technology and industry. Its versatile nature continues to drive research and innovation, paving the way for new and exciting applications in the future. The simplicity of the formula Al₂O₃ belies the depth and breadth of its impact on our world.
Latest Posts
Latest Posts
-
4 Light Years In Earth Years
Apr 03, 2025
-
27 To The Power Of 1 3
Apr 03, 2025
-
What Is The Greatest Common Factor Of 54 And 45
Apr 03, 2025
-
Finding The Gradient Of A Function
Apr 03, 2025
-
How To Find Slope Of Tangent Line At Given Point
Apr 03, 2025
Related Post
Thank you for visiting our website which covers about What Is The Correct Formula For Aluminum Oxide . We hope the information provided has been useful to you. Feel free to contact us if you have any questions or need further assistance. See you next time and don't miss to bookmark.
