What Is The Conjugate Base Of Hso3
listenit
Apr 05, 2025 · 6 min read
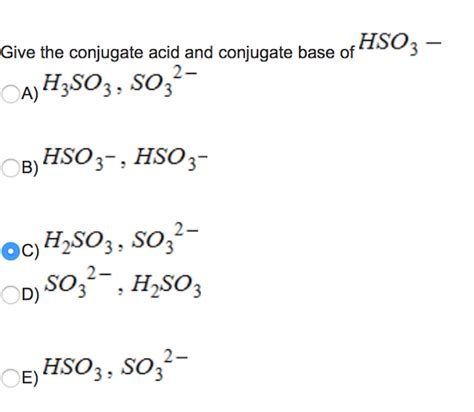
Table of Contents
What is the Conjugate Base of HSO₃⁻? Understanding Acid-Base Chemistry
Understanding conjugate acid-base pairs is fundamental to grasping acid-base chemistry. This article delves deep into the concept, focusing specifically on the conjugate base of the bisulfite ion, HSO₃⁻. We'll explore its properties, reactions, and significance in various chemical processes.
Understanding Conjugate Acid-Base Pairs
According to the Brønsted-Lowry theory, an acid is a substance that donates a proton (H⁺), while a base is a substance that accepts a proton. When an acid donates a proton, it forms its conjugate base. Conversely, when a base accepts a proton, it forms its conjugate acid. These pairs are always related by the difference of a single proton.
Think of it like a seesaw: the acid on one side donates a proton, tilting the seesaw, and the conjugate base is what remains on that side.
Example: Consider the reaction between hydrochloric acid (HCl) and water (H₂O):
HCl + H₂O ⇌ H₃O⁺ + Cl⁻
- HCl is the acid (proton donor).
- H₂O is the base (proton acceptor).
- H₃O⁺ (hydronium ion) is the conjugate acid of H₂O.
- Cl⁻ (chloride ion) is the conjugate base of HCl.
Identifying the Conjugate Base of HSO₃⁻
The bisulfite ion, HSO₃⁻, is an amphoteric species, meaning it can act as both an acid and a base. However, when considering its conjugate base, we focus on its behavior as an acid. To find the conjugate base, we simply remove a proton (H⁺) from the HSO₃⁻ ion.
The conjugate base of HSO₃⁻ is SO₃²⁻ (sulfite ion).
The reaction can be represented as:
HSO₃⁻ ⇌ H⁺ + SO₃²⁻
In this equilibrium, HSO₃⁻ acts as the acid, donating a proton to form its conjugate base, SO₃²⁻.
Properties of the Sulfite Ion (SO₃²⁻)
The sulfite ion, SO₃²⁻, is a significant species with several key properties:
1. Structure and Geometry:
SO₃²⁻ has a trigonal pyramidal geometry. The sulfur atom is at the apex, bonded to three oxygen atoms. One oxygen atom carries a formal negative charge, while the other two oxygen atoms share the remaining negative charge. This distribution contributes to its reactivity.
2. Reactivity:
-
Reducing Agent: SO₃²⁻ is a good reducing agent, meaning it readily donates electrons to other species. This is due to the presence of sulfur in a relatively low oxidation state (+4). It can be oxidized to sulfate (SO₄²⁻).
-
Base: As the conjugate base of a weak acid, SO₃²⁻ is a weak base. It can accept protons, although it's less likely to do so compared to stronger bases like hydroxide (OH⁻).
-
Nucleophile: The negative charge on the sulfite ion makes it a good nucleophile, meaning it can readily donate a lone pair of electrons to form new bonds. This property is exploited in various organic and inorganic reactions.
3. Applications:
Sulfite and bisulfite ions have wide-ranging applications, including:
-
Food Preservation: They are used as preservatives in various food and beverage products to prevent microbial growth. This is because they inhibit the growth of bacteria and fungi.
-
Pulp and Paper Industry: In the paper industry, sulfites are used in the sulfite pulping process to break down lignin, separating cellulose fibers.
-
Photography: They are used in photographic developing solutions.
-
Water Treatment: They have applications in water treatment to remove dissolved oxygen.
-
Chemical Synthesis: Sulfite ion serves as a reactant in numerous chemical synthesis processes, contributing to various important compounds.
Acid-Base Equilibrium and the Ka of HSO₃⁻
The equilibrium between HSO₃⁻ and SO₃²⁻ is governed by the acid dissociation constant (Ka) of bisulfite ion. The Ka value reflects the strength of the acid; a smaller Ka indicates a weaker acid, while a larger Ka suggests a stronger acid. The Ka value for HSO₃⁻ is relatively small, indicating it is a weak acid. This means that it doesn't readily dissociate into H⁺ and SO₃²⁻ ions in water, resulting in a relatively low concentration of H⁺ ions in solution.
The equilibrium expression is:
Ka = [H⁺][SO₃²⁻] / [HSO₃⁻]
The numerical value of Ka for HSO₃⁻ depends on temperature and ionic strength of the solution. However, it's always a relatively small value reflecting its weak acidity.
Comparison with Other Conjugate Bases
Comparing SO₃²⁻ to other conjugate bases helps illustrate its properties:
-
Compared to OH⁻ (Hydroxide): SO₃²⁻ is a much weaker base than hydroxide. Hydroxide readily accepts protons, while sulfite is a much less effective proton acceptor.
-
Compared to Cl⁻ (Chloride): Cl⁻, the conjugate base of a strong acid (HCl), is a much weaker base than SO₃²⁻. It has a negligible tendency to accept protons.
-
Compared to CH₃COO⁻ (Acetate): Acetate, the conjugate base of acetic acid, is a weak base comparable in strength to SO₃²⁻. The relative basicity of both ions would depend on the specific reaction conditions.
Significance in Chemical Reactions
The sulfite ion (SO₃²⁻) plays crucial roles in various chemical reactions:
-
Reactions with Acids: SO₃²⁻ readily reacts with acids to form bisulfite (HSO₃⁻) and ultimately sulfurous acid (H₂SO₃). This is a classic acid-base reaction.
-
Oxidation-Reduction Reactions: As mentioned earlier, SO₃²⁻ is a reducing agent. It is easily oxidized by oxidizing agents such as oxygen (O₂) or hydrogen peroxide (H₂O₂), forming sulfate (SO₄²⁻). These reactions are important in various industrial processes and environmental chemistry.
-
Addition Reactions: The nucleophilic nature of SO₃²⁻ allows it to participate in addition reactions with various electrophiles, forming new carbon-sulfur bonds. This is particularly relevant in organic chemistry.
Environmental Considerations
Sulfite and bisulfite ions have both beneficial and detrimental environmental impacts:
-
Water Pollution: High concentrations of sulfites in water bodies can be harmful to aquatic life. They can lead to oxygen depletion and impact the overall ecosystem.
-
Air Pollution: Sulfur dioxide (SO₂) emissions from industrial processes can lead to acid rain, impacting plant life and water systems. Sulfite ions are involved in the atmospheric chemistry associated with acid rain formation.
-
Food Safety: While sulfites are used as preservatives in food, they can cause allergic reactions in sensitive individuals. This highlights the importance of proper labeling and handling of food products containing sulfites.
Conclusion
The bisulfite ion (HSO₃⁻) is a crucial species in chemistry, and understanding its conjugate base, the sulfite ion (SO₃²⁻), is essential for comprehending various chemical processes. SO₃²⁻'s properties as a reducing agent, weak base, and nucleophile determine its reactivity and applications in food preservation, industrial processes, and environmental chemistry. Its role in acid-base equilibria, oxidation-reduction reactions, and addition reactions makes it a significant player in various chemical scenarios. Knowledge of its properties and behavior is crucial for anyone working in chemistry, environmental science, food science, and other related fields. Further research into the specific applications and environmental impacts of sulfite remains an important area of scientific investigation.
Latest Posts
Latest Posts
-
How Many Pounds Is A Pint
Apr 05, 2025
-
How Does Outliers Affect The Mean
Apr 05, 2025
-
What Is A Row In The Periodic Table
Apr 05, 2025
-
What Is The Square Root Of 140
Apr 05, 2025
-
How Do You Write 50 As A Decimal
Apr 05, 2025
Related Post
Thank you for visiting our website which covers about What Is The Conjugate Base Of Hso3 . We hope the information provided has been useful to you. Feel free to contact us if you have any questions or need further assistance. See you next time and don't miss to bookmark.
