What Functional Groups Are In Aspirin
listenit
Apr 02, 2025 · 6 min read
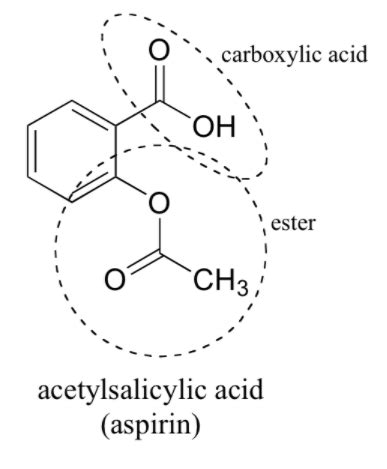
Table of Contents
What Functional Groups are in Aspirin? A Deep Dive into the Chemistry of a Common Painkiller
Aspirin, a globally recognized and widely used pain reliever and fever reducer, boasts a deceptively simple chemical structure. However, understanding the functional groups present within this seemingly unassuming molecule is crucial to grasping its pharmacological properties and how it interacts within the body. This detailed exploration will delve into the specific functional groups found in aspirin, explaining their individual roles and their collective contribution to the drug's overall effectiveness.
Understanding Functional Groups: The Building Blocks of Organic Molecules
Before diving into the specifics of aspirin, let's establish a foundational understanding of functional groups. In organic chemistry, a functional group is a specific group of atoms within a molecule that is responsible for the characteristic chemical reactions of that molecule. These groups determine the molecule's reactivity and its physical and chemical properties. They are the key to understanding how molecules interact with each other and their environment. Think of them as the building blocks that dictate a molecule's behavior.
Different functional groups impart different properties. Some are acidic, others are basic, some are reactive under certain conditions, and some are more stable. The presence and arrangement of functional groups within a molecule are paramount in determining its overall function and characteristics. Aspirin's efficacy stems directly from the specific functional groups it contains.
Deconstructing Aspirin: The Chemical Structure of Acetylsalicylic Acid
Aspirin's chemical name is acetylsalicylic acid. Its chemical formula is C₉H₈O₄. This seemingly simple formula belies a molecule containing several key functional groups that are responsible for its medicinal action. To understand these functional groups, let's examine the structural formula:
(Insert image of aspirin's chemical structure here. This image should clearly show the ester, carboxylic acid, and aromatic ring.)
The structure reveals three main functional groups:
-
Carboxylic Acid (-COOH): This is perhaps the most important functional group in aspirin. The carboxylic acid group is responsible for aspirin's acidic properties. This acidity plays a crucial role in its mechanism of action, affecting its solubility and its ability to interact with enzymes in the body. The acidic proton (H+) can be donated, leading to interactions with other molecules.
-
Ester (-COO-): The ester group is formed during the synthesis of aspirin from salicylic acid and acetic anhydride. This ester linkage is crucial to aspirin's stability. The presence of this ester, compared to the hydroxyl group in the parent compound salicylic acid, influences the properties of aspirin, such as its pharmacokinetics and its interactions within the body. It modifies the acidity compared to salicylic acid.
-
Aromatic Ring (Benzene Ring): The benzene ring is a six-carbon ring with alternating single and double bonds. This ring provides structural stability to the aspirin molecule. The aromatic ring is relatively inert compared to the other functional groups but contributes to the overall shape and interactions of the molecule, influencing how it interacts with receptors and enzymes.
The Role of Each Functional Group in Aspirin's Activity
Let's explore the specific role of each functional group in contributing to aspirin's pharmacological effects:
1. The Carboxylic Acid Group's Contribution to Aspirin's Action:
The carboxylic acid group is pivotal in aspirin's mechanism of action. It's this group that allows aspirin to inhibit cyclooxygenase (COX) enzymes, primarily COX-1 and COX-2. These enzymes are responsible for producing prostaglandins, which mediate pain, inflammation, and fever. By inhibiting COX enzymes, aspirin reduces the production of prostaglandins, leading to its analgesic (pain-relieving), anti-inflammatory, and antipyretic (fever-reducing) effects.
The acidity of the carboxylic acid group also influences aspirin's solubility and absorption in the body. The ability to donate a proton contributes to its ionization, affecting its ability to cross cell membranes and reach its target sites. This is a crucial aspect of its bioavailability.
2. The Ester Group: Stability and Modification of Salicylic Acid:
The ester group in aspirin is formed through esterification of the hydroxyl group of salicylic acid. This modification dramatically alters the properties of the parent compound. Salicylic acid itself is a potent irritant to the stomach lining and can cause significant gastrointestinal upset. The esterification that creates aspirin reduces this irritating effect by decreasing the overall acidity.
The ester linkage also contributes to the stability of aspirin. It protects the molecule from rapid hydrolysis, allowing it to remain stable under normal storage conditions. This stability ensures aspirin's effectiveness during its shelf life.
3. The Aromatic Ring: Structural Integrity and Interaction with Receptors:
While the aromatic ring is less directly involved in the primary mechanism of action, it plays a vital role in the overall structure and interactions of aspirin. Its planar nature and electronic properties influence how aspirin interacts with its target proteins and receptors, aiding in its binding to the active site of COX enzymes. The ring contributes to the overall shape and three-dimensional conformation of the molecule, influencing its interactions within the body.
Beyond the Functional Groups: Understanding Aspirin's Pharmacokinetics and Pharmacodynamics
While the functional groups are fundamental to aspirin's chemical properties, they also heavily influence its pharmacokinetics and pharmacodynamics.
Pharmacokinetics refers to how the body processes a drug, including absorption, distribution, metabolism, and excretion. The functional groups of aspirin affect how it's absorbed in the gut, distributed throughout the body, metabolized in the liver, and excreted by the kidneys. The acidic nature of the carboxylic acid group affects its solubility and absorption in the gastrointestinal tract.
Pharmacodynamics describes how the drug affects the body. Aspirin's pharmacodynamics are directly linked to the interaction between its functional groups, primarily the carboxylic acid group, and the COX enzymes. The ability to inhibit these enzymes is entirely dependent on the structural integrity and chemical properties defined by these functional groups.
Conclusion: A Complex Molecule with Simple Building Blocks
Aspirin, despite its seemingly simple molecular structure, showcases the profound impact of functional groups on a molecule's behavior. The carboxylic acid, ester, and aromatic ring each contribute uniquely to aspirin's activity, stability, and pharmacokinetics. Understanding these functional groups is not only essential for comprehending the chemical basis of aspirin's medicinal properties but also serves as a valuable example of how the interplay of functional groups within a molecule determines its overall function. This detailed understanding highlights the crucial role of functional group analysis in drug discovery and development. Further research into the specific interactions between aspirin's functional groups and its biological targets will undoubtedly lead to new advancements in pain management and the development of related medications.
Latest Posts
Latest Posts
-
What Is The Decimal Of 60
Apr 03, 2025
-
Solving Linear Systems By Substitution Answer Key
Apr 03, 2025
-
Is Radius And Diameter The Same
Apr 03, 2025
-
What Is The Symbol For Population Variance
Apr 03, 2025
-
What Is A Reactant In Photosynthesis
Apr 03, 2025
Related Post
Thank you for visiting our website which covers about What Functional Groups Are In Aspirin . We hope the information provided has been useful to you. Feel free to contact us if you have any questions or need further assistance. See you next time and don't miss to bookmark.
