What Element Has 7 Protons 8 Neutrons And 10 Electrons
listenit
Apr 06, 2025 · 4 min read
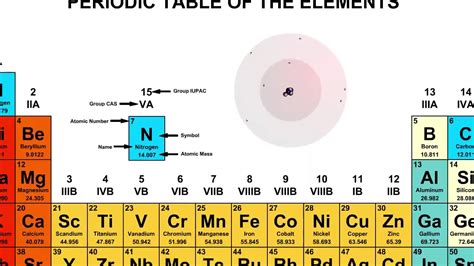
Table of Contents
What Element Has 7 Protons, 8 Neutrons, and 10 Electrons? Understanding Ions and Isotopes
The question "What element has 7 protons, 8 neutrons, and 10 electrons?" doesn't have a simple elemental answer in the traditional sense. The key to understanding this lies in the concepts of atomic number, isotopes, and ions. Let's break down each of these crucial components of atomic structure.
Atomic Number: The Defining Characteristic
The atomic number of an element is defined by the number of protons in its nucleus. This number is unique to each element and determines its place on the periodic table. Protons are positively charged subatomic particles found in the nucleus, and the number of protons dictates the element's chemical properties and behavior.
In our scenario, we have 7 protons. This immediately tells us that the element in question is nitrogen (N). Nitrogen has an atomic number of 7, meaning all nitrogen atoms contain 7 protons.
Isotopes: Variations in Neutron Number
The number of neutrons in an atom's nucleus can vary, even within the same element. These variations are called isotopes. Neutrons are neutral subatomic particles found in the nucleus alongside protons. Isotopes of the same element have the same number of protons but differ in their neutron count. This difference in neutron number doesn't alter the element's chemical properties significantly, but it can impact its nuclear stability and mass.
Our question mentions 8 neutrons. The most common isotope of nitrogen is ¹⁴N, which has 7 protons and 7 neutrons. However, an isotope of nitrogen with 8 neutrons, ¹⁵N, also exists naturally. It's a stable isotope, meaning it doesn't readily undergo radioactive decay. So, the presence of 8 neutrons is consistent with an isotope of nitrogen.
Ions: The Role of Electrons
The number of electrons in an atom determines its charge. Electrons are negatively charged subatomic particles that orbit the nucleus. In a neutral atom, the number of electrons equals the number of protons, resulting in a net charge of zero. However, atoms can gain or lose electrons, forming ions.
- Cations: When an atom loses electrons, it becomes positively charged, forming a cation.
- Anions: When an atom gains electrons, it becomes negatively charged, forming an anion.
Our question specifies 10 electrons. Since nitrogen has 7 protons, the presence of 10 electrons indicates that the nitrogen atom has gained 3 electrons (10 - 7 = 3). This means we're dealing with a nitrogen anion, specifically a nitride ion (N³⁻).
Bringing It All Together: The Answer
Therefore, the answer to the question, "What element has 7 protons, 8 neutrons, and 10 electrons?" is a nitride ion (N³⁻) derived from the isotope ¹⁵N. It's crucial to understand that it's not a neutral nitrogen atom but rather an ion with a -3 charge due to the excess of three electrons.
Deeper Dive into Isotopes and Their Significance
The existence of isotopes is a fundamental concept in chemistry and nuclear physics. Different isotopes of an element can have varying abundances in nature. For instance, ¹⁴N is far more abundant than ¹⁵N. The relative abundance of isotopes is often expressed as a percentage.
The study of isotopes has numerous applications:
-
Radioactive Isotopes in Medicine: Radioactive isotopes, which are unstable and undergo decay, are used in medical imaging techniques like PET (positron emission tomography) scans and radiotherapy for cancer treatment. These isotopes emit radiation that can be detected and used to diagnose and treat diseases.
-
Radioactive Dating: Radioactive isotopes with known half-lives (the time it takes for half of the isotope to decay) are used in radiometric dating to determine the age of rocks, fossils, and other materials. Carbon-14 dating, which uses the radioactive isotope ¹⁴C, is a prime example of this application.
-
Tracing Chemical Processes: Stable isotopes can be used as tracers to follow the movement of atoms and molecules in chemical reactions and biological processes. This technique is used in various fields, including environmental science and biochemistry.
-
Nuclear Energy: Certain isotopes, like uranium-235, are used as fuel in nuclear power plants. Nuclear fission of these isotopes releases a tremendous amount of energy.
Exploring the Properties of Nitride Ions
Nitride ions (N³⁻) are important in various chemical compounds. They are typically formed when nitrogen reacts with highly electropositive metals, such as alkali and alkaline earth metals. Nitrides exhibit diverse properties depending on the metal they're bonded with. Some nitrides are hard and refractory materials with high melting points, making them suitable for applications in high-temperature environments.
The reactivity of nitride ions is largely influenced by the electronegativity difference between nitrogen and the metal it's bonded with. The higher the electronegativity difference, the more ionic the nitride compound will be.
Conclusion: A Multifaceted Answer
The initial question seems straightforward, but it reveals the complexity and richness of atomic structure. The answer – a nitride ion (N³⁻) derived from the isotope ¹⁵N – highlights the critical interplay of protons, neutrons, and electrons in determining an atom's identity and properties. Understanding these fundamental concepts is essential for comprehending the behavior of matter and its applications across various scientific disciplines. The exploration of isotopes and their significance further underscores the multifaceted nature of atomic structure and its far-reaching implications.
Latest Posts
Latest Posts
-
Best Point Estimate Of The Population Mean
Apr 06, 2025
-
Does Ionization Energy Increase Across A Period
Apr 06, 2025
-
What Is 1 66666 As A Fraction
Apr 06, 2025
-
What Is The Si Base Unit For Length
Apr 06, 2025
-
93 Is What Percent Of 186
Apr 06, 2025
Related Post
Thank you for visiting our website which covers about What Element Has 7 Protons 8 Neutrons And 10 Electrons . We hope the information provided has been useful to you. Feel free to contact us if you have any questions or need further assistance. See you next time and don't miss to bookmark.
