What Are Three Parts Of Atp Molecule
listenit
Apr 01, 2025 · 6 min read
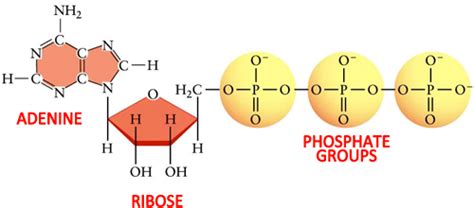
Table of Contents
What are the Three Parts of an ATP Molecule? A Deep Dive into Adenosine Triphosphate
Adenosine triphosphate (ATP) is often called the "energy currency" of life. This small but mighty molecule fuels countless cellular processes, from muscle contraction and nerve impulse transmission to protein synthesis and DNA replication. Understanding its structure is crucial to comprehending its function. This article delves deep into the three constituent parts of the ATP molecule, exploring their individual roles and how they contribute to ATP's remarkable ability to store and release energy.
The Three Key Components of ATP: A Detailed Look
The ATP molecule is composed of three distinct parts:
- Adenine: A nitrogenous base.
- Ribose: A five-carbon sugar.
- Triphosphate: A chain of three phosphate groups.
Let's examine each component in detail:
1. Adenine: The Nitrogenous Base Foundation
Adenine is a purine base, a type of nitrogen-containing molecule with a double-ring structure. It's one of the fundamental building blocks of nucleic acids, DNA and RNA, where it pairs with thymine (in DNA) or uracil (in RNA). In ATP, adenine's role is primarily structural. Its planar aromatic ring system contributes to the overall stability of the molecule and participates in weak interactions with other molecules within the cell.
Key Characteristics of Adenine in ATP:
- Planar Structure: The flat structure of adenine allows for efficient stacking interactions with other molecules.
- Nitrogenous Nature: The nitrogen atoms in adenine can participate in hydrogen bonding, contributing to the molecule's interactions with other cellular components.
- Hydrophobic Properties: Parts of the adenine ring exhibit hydrophobic (water-repelling) properties, influencing its interactions within the hydrophobic core of proteins or membranes.
The specific arrangement of adenine within the ATP molecule is crucial. It's attached to the ribose sugar via a glycosidic bond, a stable covalent link that ensures adenine remains firmly connected to the rest of the molecule. This strong bond prevents spontaneous detachment, ensuring the integrity of the ATP molecule under various cellular conditions.
2. Ribose: The Sugar Backbone
Ribose is a five-carbon sugar, a pentose, forming the backbone of the ATP molecule. It's a crucial structural component, connecting adenine to the triphosphate group. The specific isomer of ribose found in ATP is β-D-ribose, meaning it has a specific spatial arrangement of its hydroxyl groups.
Key Characteristics of Ribose in ATP:
- Five-Carbon Sugar: The five-carbon structure provides the necessary framework for connecting adenine and the triphosphate group.
- Hydroxyl Groups: The hydroxyl groups on ribose participate in hydrogen bonding and contribute to the molecule's solubility in water. Their specific positions influence the three-dimensional shape and reactivity of the molecule.
- β-D-Ribose Configuration: This specific configuration is essential for ATP's function and its interaction with enzymes.
The ribose sugar is connected to adenine via a N-glycosidic bond and to the phosphate group via an ester bond. These bonds are crucial for maintaining the structural integrity of the ATP molecule. The specific conformation of the ribose sugar also plays a role in enzyme recognition and binding, influencing the efficiency of ATP utilization in metabolic reactions.
3. Triphosphate: The Energy-Rich Tail
The triphosphate group is arguably the most crucial part of the ATP molecule. This tail consists of three phosphate groups linked together by high-energy phosphoanhydride bonds. It's these bonds that hold the key to ATP's energy-storing capabilities.
Key Characteristics of the Triphosphate Group:
- High-Energy Phosphoanhydride Bonds: The bonds between the phosphate groups are particularly high-energy, meaning they release a significant amount of energy when broken. This is due to the electrostatic repulsion between the negatively charged phosphate groups. The proximity of these negatively charged groups creates a state of high potential energy.
- Negative Charge: The multiple negative charges on the phosphate groups contribute to the molecule's high potential energy and influence its interactions with enzymes and other molecules.
- Hydrolysis: The hydrolysis of these bonds, where a water molecule is used to break a phosphoanhydride bond, releases a significant amount of free energy. This energy is then harnessed by the cell to drive various metabolic processes.
The energy released during ATP hydrolysis is not simply heat; it's used to drive endergonic reactions—reactions that require energy input. This is achieved through several mechanisms, often involving the transfer of a phosphate group from ATP to another molecule, thereby altering the energy state of the recipient molecule and allowing it to undergo otherwise unfavorable reactions.
ATP Hydrolysis: The Engine of Cellular Work
The process of ATP hydrolysis is central to the molecule's role as the energy currency of the cell. It's the breaking of the phosphoanhydride bond between the second and third phosphate groups (yielding ADP and inorganic phosphate, Pi), which releases the energy that powers cellular processes. This process is highly regulated by various enzymes, ensuring that energy is released in a controlled and efficient manner.
The Mechanism of ATP Hydrolysis:
- Enzyme Binding: ATP binds to a specific enzyme involved in the process requiring energy.
- Hydrolysis: A water molecule reacts with the phosphoanhydride bond, breaking it and releasing inorganic phosphate (Pi) and ADP (adenosine diphosphate).
- Energy Transfer: The energy released during bond breakage is utilized to drive the endergonic reaction, often through conformational changes in the enzyme or the transfer of a phosphate group to another molecule.
- Product Release: The ADP and Pi are released, allowing the enzyme to bind another ATP molecule and repeat the cycle.
It's important to note that ATP hydrolysis doesn't directly power all cellular processes. Often, the energy released is used to create transient high-energy intermediates, which then drive the specific reaction. This mechanism allows for a controlled and efficient utilization of energy.
ATP Regeneration: The Continuous Cycle
While ATP hydrolysis releases energy, the cell needs a continuous supply of ATP to function. This is achieved through ATP regeneration, a process where ADP and Pi are converted back into ATP. The primary methods of ATP regeneration include:
- Cellular Respiration: This is the most important pathway, involving the breakdown of glucose and other fuel molecules to generate ATP. It occurs in the mitochondria and involves a series of complex biochemical reactions, including glycolysis, the Krebs cycle, and oxidative phosphorylation.
- Photosynthesis: In plants and other photosynthetic organisms, ATP is generated through the light-dependent reactions of photosynthesis. Light energy is used to drive the synthesis of ATP from ADP and Pi.
- Substrate-Level Phosphorylation: In some metabolic pathways, ATP is generated directly through the transfer of a phosphate group from a high-energy substrate molecule to ADP. This method is less efficient than cellular respiration or photosynthesis but plays a vital role in certain metabolic processes.
Conclusion: ATP, the Universal Energy Currency
The ATP molecule, with its three crucial components—adenine, ribose, and triphosphate—is fundamental to life. Its unique structure, particularly the high-energy phosphoanhydride bonds in the triphosphate tail, allows it to store and release energy efficiently. The continuous cycle of ATP hydrolysis and regeneration ensures a constant supply of energy to fuel the diverse processes that sustain life. A deeper understanding of ATP’s structure provides crucial insight into the intricate mechanisms that drive cellular activity and maintain life's delicate balance. Further research continues to unravel the complexities of ATP metabolism and its critical role in health and disease.
Latest Posts
Latest Posts
-
What Is 5 6 As A Decimal
Apr 03, 2025
-
The Conjugate Base Of Hso4 Is
Apr 03, 2025
-
Examples Of Gay Lussacs Law In Real Life
Apr 03, 2025
-
Law Of Sines And Law Of Cosines Word Problems
Apr 03, 2025
-
18 Is What Percent Of 36
Apr 03, 2025
Related Post
Thank you for visiting our website which covers about What Are Three Parts Of Atp Molecule . We hope the information provided has been useful to you. Feel free to contact us if you have any questions or need further assistance. See you next time and don't miss to bookmark.
