Examples Of Gay Lussac's Law In Real Life
listenit
Apr 03, 2025 · 7 min read
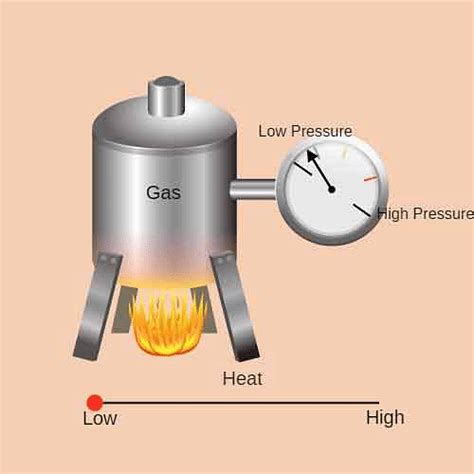
Table of Contents
Examples of Gay-Lussac's Law in Real Life: From Pressure Cookers to Balloons
Gay-Lussac's Law, also known as Amonton's Law, is a fundamental gas law that describes the relationship between the pressure and temperature of a gas when the volume is held constant. It states that the pressure of a gas is directly proportional to its absolute temperature, provided that the volume and amount of gas remain constant. This means that if you increase the temperature of a gas, its pressure will also increase, and vice versa. While it might seem like a theoretical concept confined to textbooks, Gay-Lussac's Law is actively at play in numerous real-life scenarios. Understanding this law is crucial for safety, efficiency, and even enjoying everyday activities. Let's explore several compelling examples:
Everyday Applications of Gay-Lussac's Law
1. Pressure Cookers: Perhaps one of the most common and readily understandable examples of Gay-Lussac's Law in action is the humble pressure cooker. Pressure cookers work by trapping steam inside a sealed container. As the water boils, the temperature increases, and consequently, the pressure inside the cooker rises significantly. This elevated pressure leads to higher temperatures than are achievable in standard open-air cooking, resulting in faster cooking times and more tender food. The safety valve on the pressure cooker is a critical component, designed to release pressure and prevent potentially dangerous explosions if the temperature and pressure become excessively high.
Understanding the safety implications: Ignoring the safety mechanisms of a pressure cooker can have dire consequences. If the pressure builds up beyond the cooker's capacity, a catastrophic failure can occur, leading to hot, pressurized contents being released violently. This underscores the importance of understanding and respecting the principles of Gay-Lussac's Law in everyday kitchen appliances.
2. Aerosol Cans: Aerosol cans, from hairspray to whipped cream, utilize Gay-Lussac's Law to their advantage. These cans contain a propellant gas under pressure, often a liquefied gas. When the valve is depressed, this compressed gas expands rapidly, pushing the product out. As the gas expands, its temperature drops according to another gas law (Joule-Thomson effect), causing a slight cooling effect on the can. The initial pressure of the gas is directly related to its temperature within the sealed can. Changes in ambient temperature will affect the internal pressure of the aerosol can – hotter temperatures lead to higher internal pressures.
Safety Considerations for Aerosol Cans: Never expose aerosol cans to excessive heat, like leaving them in direct sunlight or near a flame. High temperatures cause a proportional increase in pressure, potentially leading to rupture and the release of the contents.
3. Hot Air Balloons: The principle behind hot air balloons perfectly demonstrates Gay-Lussac's Law. By heating the air inside the balloon envelope, the air expands and becomes less dense. Because the volume of the balloon remains relatively constant, the increase in temperature directly leads to a proportional increase in air pressure. This increased pressure then lifts the balloon. The pilot controls the altitude of the balloon by adjusting the temperature of the air inside the envelope – increasing the temperature causes it to rise, and decreasing the temperature causes it to descend.
Understanding the delicate balance: The successful operation of a hot air balloon relies on a precise balance between the heated air inside and the cooler surrounding air. The difference in pressure, created by the temperature difference, provides the lift.
4. Tires: The pressure in car or bicycle tires is another excellent example of Gay-Lussac's Law. On hot summer days, the temperature of the air inside the tires increases. This increase in temperature leads to a corresponding increase in pressure. This is why it's crucial to check tire pressure regularly, particularly during extreme temperature changes. Over-inflation can cause tire damage, while under-inflation reduces fuel efficiency and handling.
Avoiding potential dangers: Over-inflated tires are more susceptible to bursting, while under-inflated tires can overheat and increase the risk of a blowout. Regular tire pressure monitoring helps maintain optimal pressure and ensures safety.
Less Obvious Applications and Scientific Implications
5. Internal Combustion Engines: Internal combustion engines in vehicles and power generators rely on the principles of Gay-Lussac's Law. The controlled explosions of fuel within the cylinders generate significant heat, which translates to a rapid increase in pressure. This pressure pushes the pistons, converting the chemical energy into mechanical energy. Efficient engine operation depends critically on the precise control and management of pressure and temperature within the engine cylinders.
The complex dance of combustion: The timing and control of the combustion process is crucial. The interaction of temperature and pressure during each cycle drives the engine's performance and efficiency.
6. Weather Patterns: While more complex than a simple application of Gay-Lussac's Law, atmospheric pressure changes are partly explained by temperature variations. Warmer air masses tend to have higher pressure than colder air masses, contributing to weather patterns like high- and low-pressure systems. This temperature-pressure relationship is a fundamental aspect of meteorology and weather forecasting.
The larger picture: While many other factors influence weather patterns, temperature gradients and the consequent pressure variations play a significant role in atmospheric dynamics.
7. Scuba Diving: Scuba divers must understand the effects of pressure changes with depth. As divers descend, the water pressure increases. This increased pressure affects the volume of gases within their bodies, including their lungs. The increased pressure compresses the air in their tanks, thus affecting how long they can stay underwater and influencing the rate at which they need to ascend.
Safety is paramount: Ignoring the effects of pressure at various depths during scuba diving is highly dangerous and could lead to serious injury or death due to gas expansion or compression within the body.
8. Industrial Processes: Many industrial processes involve gases at high temperatures and pressures. Chemical reactors, refineries, and power plants rely on a thorough understanding of Gay-Lussac's Law to ensure safety and efficiency. Accurate calculations based on this law are crucial for designing and operating these processes safely and effectively.
The importance of precision: In industrial settings, the slightest miscalculation can have significant safety and economic consequences. Adherence to the principles of Gay-Lussac's Law is essential for optimized performance and minimizing risk.
Beyond the Basics: Further Considerations
Understanding limitations: While Gay-Lussac's Law is a powerful tool, it's important to remember that it's an ideal gas law. Real-world gases don't always behave exactly according to the ideal model. At very high pressures or low temperatures, deviations from the ideal gas law can occur. In such situations, more complex equations of state are necessary for accurate calculations.
Combining gas laws: Gay-Lussac's Law is often used in conjunction with other gas laws, such as Boyle's Law and Charles's Law, to solve more complex problems involving changing conditions of temperature, pressure, and volume. The combined gas law consolidates these principles into a single equation.
Real-world applications demand context: The examples above illustrate how Gay-Lussac's Law is prevalent in diverse real-life applications. However, the law’s impact is rarely isolated; it frequently interacts with other physical phenomena, like the Joule-Thomson effect in aerosols or the complex thermodynamics of an internal combustion engine. This interdependency is vital to consider when analysing these systems.
Further exploration: The understanding of Gay-Lussac’s Law fosters a deeper appreciation for the principles governing gas behavior. This foundational knowledge opens doors to further exploration of related concepts in physics, chemistry, and engineering, which ultimately contribute to technological advancements and our ability to safely and efficiently interact with the world around us.
In conclusion, Gay-Lussac's Law is far from a mere theoretical concept. It underpins numerous crucial aspects of our daily lives and various industrial processes. From the simple act of cooking to the complex workings of internal combustion engines, this fundamental gas law plays a vital role, highlighting its practical significance and far-reaching influence. Understanding this law isn't just about passing a physics exam; it's about comprehending the underlying principles that govern many facets of our modern world.
Latest Posts
Latest Posts
-
What Is The Square Root Of 3 2
Apr 03, 2025
-
What Quadrilateral Is Not A Parallelogram
Apr 03, 2025
-
What Is The Decimal For 4 6
Apr 03, 2025
-
What Is 14 Inches In Centimeters
Apr 03, 2025
-
How To Make An Exponent Positive
Apr 03, 2025
Related Post
Thank you for visiting our website which covers about Examples Of Gay Lussac's Law In Real Life . We hope the information provided has been useful to you. Feel free to contact us if you have any questions or need further assistance. See you next time and don't miss to bookmark.
