The Si Unit For Energy Is
listenit
Apr 02, 2025 · 5 min read
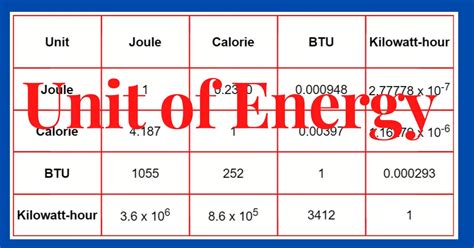
Table of Contents
The SI Unit for Energy Is: A Deep Dive into the Joule
The SI unit for energy is the joule (J). This seemingly simple statement belies a rich history and a profound impact across numerous scientific disciplines. Understanding the joule, its implications, and its relationship to other units is crucial for anyone working with energy, from physicists exploring fundamental forces to engineers designing efficient power systems. This article delves deep into the joule, exploring its definition, applications, conversions, and significance in various fields.
Defining the Joule: Work, Energy, and Power
The joule is defined as the work done when a force of one newton (N) is applied over a displacement of one meter (m). This seemingly straightforward definition encapsulates a fundamental concept in physics: the relationship between work, energy, and power.
-
Work: Work is done when a force causes an object to move a certain distance. It's a measure of energy transfer. The formula for work (W) is:
W = Fd, where 'F' is the force in newtons and 'd' is the displacement in meters. -
Energy: Energy is the capacity to do work. The joule, therefore, is a unit not just of work, but also of energy in all its forms, including kinetic energy (energy of motion), potential energy (stored energy), thermal energy (heat), chemical energy, and nuclear energy.
-
Power: Power is the rate at which work is done or energy is transferred. It's measured in watts (W), where one watt is equal to one joule per second (J/s). A higher power rating indicates a faster energy transfer.
Therefore, the joule provides a unifying measure for different energy forms and their transfer. This consistency is a key advantage in scientific analysis and engineering design.
Applications of the Joule Across Disciplines
The joule's versatility as a unit of energy makes it indispensable across a wide spectrum of fields:
1. Physics: From Classical Mechanics to Quantum Physics
In classical mechanics, the joule is used extensively to calculate work, kinetic energy, and potential energy. For example, the kinetic energy (KE) of an object is given by: KE = 1/2mv², where 'm' is the mass in kilograms and 'v' is the velocity in meters per second. The result is expressed in joules.
In quantum physics, even though energy is often expressed in electronvolts (eV), the joule serves as a fundamental unit, underlying the conversions and calculations involving energy levels of atoms and molecules.
2. Engineering: Power Generation, Transmission, and Consumption
Engineers use the joule to design and analyze energy systems, from power plants generating electricity to the consumption of energy by appliances. The efficiency of a system is often expressed as the ratio of useful energy output (in joules) to the energy input.
The joule is essential in calculating energy losses in transmission lines and optimizing energy distribution networks. Understanding energy consumption in joules is crucial for developing energy-efficient technologies and reducing environmental impact.
3. Chemistry: Chemical Reactions and Thermodynamics
In chemistry, the joule is used to quantify the energy changes that occur during chemical reactions. Enthalpy changes (ΔH), which represent the heat absorbed or released during a reaction at constant pressure, are measured in joules. The joule is also crucial in understanding thermodynamic concepts like Gibbs free energy, which predicts the spontaneity of a reaction.
4. Meteorology: Weather Systems and Energy Transfer
Meteorology utilizes the joule to measure the energy involved in atmospheric processes, such as the formation of clouds, thunderstorms, and hurricanes. The energy transferred during phase changes (like evaporation and condensation) is quantified in joules. Understanding these energy transfers is vital for weather forecasting and climate modeling.
5. Biology: Biological Processes and Metabolism
In biology, the joule finds application in measuring the energy released during metabolic processes. The energy content of food is often expressed in joules, reflecting the energy available for biological work. The joule helps quantify the energy expenditure during physical activity, contributing to understanding the relationship between diet and exercise.
Conversions and Other Related Units
While the joule is the primary SI unit for energy, other units are commonly used, often based on practical considerations or historical context. Understanding the conversions is crucial for seamless integration across different fields.
-
Kilojoule (kJ): 1 kJ = 1000 J. This is often used for larger energy quantities, such as the energy content of food or the energy output of a power plant.
-
Megajoule (MJ): 1 MJ = 1,000,000 J. Used for even larger energy values.
-
Gigajoule (GJ): 1 GJ = 1,000,000,000 J. Commonly used in large-scale energy applications.
-
Calorie (cal) and Kilocalorie (kcal): These units are often used in nutritional contexts. 1 kcal (also known as a Calorie, with a capital C) is approximately equal to 4184 J. Conversions are essential to bridge the gap between scientific and everyday language regarding energy.
-
Electronvolt (eV): This unit is primarily used in atomic and nuclear physics. 1 eV is approximately equal to 1.602 x 10⁻¹⁹ J. It's useful for expressing the energy of individual particles.
-
British Thermal Unit (BTU): A unit commonly used in the United States for heating and cooling systems. 1 BTU is approximately equal to 1055 J.
The Joule and the Future of Energy
The joule will continue to play a critical role in shaping our understanding and management of energy. As we move towards a more sustainable energy future, accurate energy measurement and conversion are crucial. The joule, as the fundamental SI unit, provides the necessary foundation for research, development, and innovation in renewable energy technologies, energy efficiency, and sustainable energy policies.
Conclusion
The joule, seemingly a simple unit, is a powerful tool that underpins our understanding of energy across diverse scientific and engineering disciplines. Its consistent application ensures seamless integration of energy-related data, calculations, and analyses, promoting effective communication and collaboration. From fundamental physics to large-scale energy systems, the joule remains the cornerstone of energy quantification, paving the way for advancements in energy technology and sustainable practices. The widespread acceptance and consistent application of the joule cement its crucial role in the ever-evolving landscape of energy. Understanding the joule is key to comprehending the world around us and shaping a more energy-efficient future.
Latest Posts
Latest Posts
-
How Are Elements Arranged In The Modern Periodic Table
Apr 03, 2025
-
How To Integrate On A Ti 84
Apr 03, 2025
-
Two Lines Intersecting At A Right Angle
Apr 03, 2025
-
What Is 7 Out Of 15 As A Percentage
Apr 03, 2025
-
Which Kingdom Contains Heterotrophs With Cell Walls Of Chitin
Apr 03, 2025
Related Post
Thank you for visiting our website which covers about The Si Unit For Energy Is . We hope the information provided has been useful to you. Feel free to contact us if you have any questions or need further assistance. See you next time and don't miss to bookmark.
