Oxidation State Of Cr In Cr2o72
listenit
Apr 05, 2025 · 5 min read
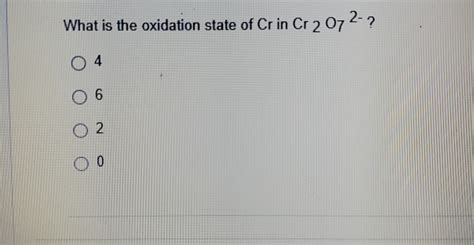
Table of Contents
Delving Deep into the Oxidation State of Cr in Cr₂O₇²⁻
The dichromate anion, Cr₂O₇²⁻, is a vibrant orange species frequently encountered in chemistry, particularly in redox reactions. Understanding its structure and, crucially, the oxidation state of chromium (Cr) within the anion, is fundamental to predicting its reactivity and applications. This article will comprehensively explore the oxidation state of Cr in Cr₂O₇²⁻, delving into the intricacies of its determination and its implications.
Determining the Oxidation State of Chromium in Dichromate
The oxidation state, or oxidation number, of an atom represents the hypothetical charge it would possess if all bonds to atoms of different elements were 100% ionic. This concept is crucial for balancing redox reactions and understanding the chemical behavior of compounds. To determine the oxidation state of chromium in Cr₂O₇²⁻, we utilize the following steps:
Step 1: Assign Oxidation States to Known Elements
We start by assigning oxidation states to elements with consistently predictable oxidation numbers. In Cr₂O₇²⁻, oxygen (O) almost always exhibits an oxidation state of -2, except in peroxides and other unusual compounds.
Step 2: Set up an Algebraic Equation
Let 'x' represent the oxidation state of chromium (Cr). Since there are two chromium atoms in the dichromate ion, the total contribution from chromium is 2x. There are seven oxygen atoms, each contributing -2, resulting in a total contribution of 7(-2) = -14. The overall charge of the dichromate ion is -2. Therefore, we can write the equation:
2x + 7(-2) = -2
Step 3: Solve for the Oxidation State of Chromium
Solving the equation for 'x':
2x - 14 = -2 2x = 12 x = +6
Therefore, the oxidation state of chromium (Cr) in Cr₂O₇²⁻ is +6.
The Significance of the +6 Oxidation State of Chromium
The +6 oxidation state is the highest oxidation state that chromium can achieve. This high oxidation state makes chromium(VI) compounds, such as dichromate, strong oxidizing agents. This is because chromium in the +6 state has a strong tendency to gain electrons and reduce to lower oxidation states, such as +3.
Implications for Redox Reactions
The strong oxidizing power of Cr₂O₇²⁻ is exploited in numerous applications, including:
-
Organic Chemistry: Dichromate is used as an oxidizing agent in various organic reactions, such as the oxidation of alcohols to aldehydes or ketones. The strong oxidizing power allows for the efficient conversion of functional groups.
-
Analytical Chemistry: Dichromate is employed in titrations to determine the concentration of reducing agents. The color change from orange (Cr₂O₇²⁻) to green (Cr³⁺) serves as a visual indicator of the endpoint.
-
Industrial Applications: Dichromate has been used in various industrial processes, including leather tanning, metal finishing, and wood preservation. However, due to its toxicity and environmental concerns, its use has been significantly curtailed.
Structural Aspects and Oxidation State
The structure of the dichromate ion also plays a role in understanding the +6 oxidation state of chromium. The ion consists of two chromium atoms each bonded to four oxygen atoms, with one oxygen atom bridging the two chromium atoms. This bridging oxygen atom contributes to the overall stability of the +6 oxidation state. The strong Cr=O bonds help stabilize the high oxidation state of chromium.
Comparison with Chromate (CrO₄²⁻)
It's helpful to compare the dichromate ion with the chromate ion (CrO₄²⁻). In chromate, the oxidation state of chromium is also +6. However, the chromate ion is a tetrahedral structure, while dichromate is formed by two chromate units linked by a shared oxygen atom. Both ions possess similar oxidizing properties, although the relative strength might slightly vary due to differences in their structures.
Environmental Concerns and Toxicity of Chromium(VI)
It's crucial to acknowledge the significant environmental and health concerns associated with chromium(VI) compounds. Chromium(VI) is classified as a human carcinogen and poses a severe threat to both human health and the environment. Its high toxicity necessitates careful handling and disposal. The widespread use of chromium(VI) compounds in the past has led to significant environmental contamination.
Sustainable Alternatives
Due to the toxicity of chromium(VI), researchers are actively exploring more sustainable and environmentally friendly alternatives for applications previously reliant on dichromate. These include exploring less toxic oxidizing agents and developing cleaner industrial processes.
Detailed Analysis of Redox Reactions Involving Cr₂O₇²⁻
Understanding the redox reactions involving dichromate requires a clear grasp of the electron transfer processes involved. When Cr₂O₇²⁻ acts as an oxidizing agent, it undergoes a reduction, with chromium's oxidation state decreasing from +6 to a lower state, typically +3.
Balancing Redox Reactions with Dichromate
Balancing redox reactions with dichromate often involves the half-reaction method. This involves separating the overall reaction into two half-reactions: one for oxidation and one for reduction.
Example: Oxidation of Fe²⁺ by Cr₂O₇²⁻ in acidic medium:
The balanced half-reactions are:
Reduction: Cr₂O₇²⁻ + 14H⁺ + 6e⁻ → 2Cr³⁺ + 7H₂O
Oxidation: 6Fe²⁺ → 6Fe³⁺ + 6e⁻
By combining these half-reactions and canceling out the electrons, we obtain the balanced overall redox reaction:
Cr₂O₇²⁻ + 14H⁺ + 6Fe²⁺ → 2Cr³⁺ + 6Fe³⁺ + 7H₂O
This balanced equation clearly illustrates the electron transfer process, with six electrons being transferred from six Fe²⁺ ions to one Cr₂O₇²⁻ ion.
Applications Beyond Redox Chemistry
While the strong oxidizing properties of Cr₂O₇²⁻ dominate its applications, it also finds use in other areas. For example, certain chromium(VI) compounds display specific optical properties that are relevant to some technological applications.
Future Research Directions
Research continues to explore the chemistry of chromium(VI) compounds, focusing on:
-
Developing safer and more sustainable alternatives: This research aims to replace Cr(VI) compounds with less toxic alternatives in industrial processes.
-
Understanding the environmental fate and transport of chromium(VI): This research helps to assess and mitigate the environmental impacts of chromium(VI) contamination.
-
Exploring novel applications of chromium(VI) compounds in specific niches: This research might involve exploiting unique optical or catalytic properties.
Conclusion
The oxidation state of chromium in Cr₂O₇²⁻ is unequivocally +6. This high oxidation state is responsible for the compound's strong oxidizing properties, which are widely exploited in various applications, ranging from organic chemistry to analytical chemistry and various industrial processes. However, the inherent toxicity and environmental concerns associated with chromium(VI) necessitate the development and implementation of safer alternatives. Understanding the oxidation state and its implications is key to both appreciating the utility and addressing the challenges associated with this significant chemical species. Further research is essential to develop sustainable solutions and mitigate the risks associated with the use of chromium(VI) compounds.
Latest Posts
Latest Posts
-
What Is The Square Root Of 2 25
Apr 05, 2025
-
What Is The Least Common Multiple Of 9 And 3
Apr 05, 2025
-
The Horizontal Columns On The Periodic Table Are Called
Apr 05, 2025
-
What Is The Least Common Multiple Of 14 And 7
Apr 05, 2025
-
7 Cm Is How Many Inches
Apr 05, 2025
Related Post
Thank you for visiting our website which covers about Oxidation State Of Cr In Cr2o72 . We hope the information provided has been useful to you. Feel free to contact us if you have any questions or need further assistance. See you next time and don't miss to bookmark.
