Number Of Valence Electrons For Carbon
listenit
Apr 02, 2025 · 6 min read
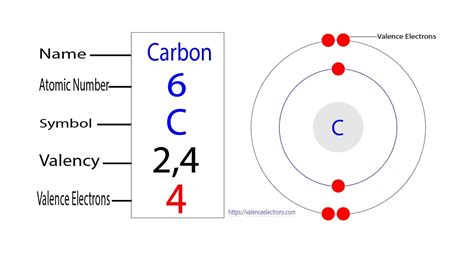
Table of Contents
The Significance of Valence Electrons: A Deep Dive into Carbon's Four
Carbon, the backbone of life and a cornerstone of organic chemistry, owes its remarkable versatility to a seemingly simple feature: its four valence electrons. Understanding the number and behavior of these electrons is crucial to comprehending carbon's unparalleled ability to form diverse and complex molecules. This article delves deep into the world of carbon's valence electrons, exploring their significance in bonding, the implications for molecular structure and properties, and the broader implications for the fields of chemistry and materials science.
What are Valence Electrons?
Before we focus specifically on carbon, let's establish a foundational understanding of valence electrons. Valence electrons are the electrons located in the outermost shell, or energy level, of an atom. These electrons are the most loosely held and, therefore, are primarily responsible for the atom's chemical behavior and its ability to form chemical bonds with other atoms. The number of valence electrons determines an element's reactivity and the types of bonds it can form – single, double, or triple bonds. This number is directly related to an element's position within the periodic table, specifically its group number.
Carbon's Unique Position: Group 14
Carbon resides in Group 14 (or IVA) of the periodic table. This group placement is pivotal because it dictates that carbon atoms possess four valence electrons. This seemingly small number is the driving force behind carbon's exceptional ability to form a vast array of molecules. Unlike elements with one or two valence electrons that primarily form simpler structures, carbon's four valence electrons enable it to create complex, branched, and ringed structures that underpin the diversity of organic chemistry.
The Significance of Carbon's Four Valence Electrons
Carbon's four valence electrons have several crucial implications:
1. Tetrahedral Bonding Geometry
The most common bonding configuration for carbon involves sharing its four valence electrons to form four covalent bonds. This results in a tetrahedral geometry, with bond angles of approximately 109.5 degrees. This geometrical arrangement is fundamental to the three-dimensional structure of many organic molecules, influencing their shape, properties, and interactions.
2. Formation of Long Chains and Rings
The ability of carbon to form stable bonds with other carbon atoms is extraordinary. This property allows carbon to create long chains, branched chains, and ring structures. This chain-forming capacity is unparalleled among other elements and is the basis for the existence of polymers, long molecules composed of repeating subunits. Examples abound: from simple hydrocarbons like propane to complex biomolecules like DNA.
3. Diverse Bond Types
Carbon's four valence electrons permit the formation of various bond types, including:
- Single bonds: A single covalent bond involves the sharing of one pair of electrons between two carbon atoms (or a carbon atom and another atom).
- Double bonds: Two pairs of electrons are shared between two carbon atoms, resulting in a stronger and shorter bond than a single bond. This is crucial in molecules like alkenes and carbon dioxide.
- Triple bonds: Three pairs of electrons are shared between two carbon atoms, resulting in the strongest and shortest bond type. This is characteristic of alkynes, like acetylene.
The ability to form these different bond types significantly increases the structural diversity of carbon-containing molecules.
4. Isomerism
The multiplicity of bonding possibilities also leads to the phenomenon of isomerism. Isomers are molecules with the same molecular formula but different structural arrangements. This diversity arises directly from carbon's ability to form single, double, and triple bonds and arrange itself into linear, branched, or cyclic structures. The existence of isomers vastly expands the number of possible carbon-based compounds.
5. Carbon's Role in Biological Systems
The remarkable properties stemming from carbon's four valence electrons are essential for life as we know it. Carbon's ability to form long chains and complex structures allows the formation of:
- Carbohydrates: Energy sources and structural components.
- Lipids: Essential for cell membranes and energy storage.
- Proteins: The workhorses of cells, performing diverse functions.
- Nucleic acids (DNA and RNA): The carriers of genetic information.
Without carbon's unique bonding capabilities, the complexity and diversity of life would be unimaginable.
Beyond Organic Chemistry: Carbon in Materials Science
The impact of carbon's four valence electrons extends far beyond the realm of organic chemistry. Various allotropes of carbon, each with unique structural arrangements and properties, highlight this versatility:
-
Diamond: A three-dimensional network of carbon atoms bonded through strong covalent bonds, resulting in exceptional hardness and refractive index. The tetrahedral arrangement is crucial here.
-
Graphite: A layered structure where carbon atoms are arranged in hexagonal lattices. The weak interactions between the layers account for graphite's softness and ability to act as a lubricant. Each carbon atom is still bonded to three others within its layer.
-
Fullerenes (e.g., buckminsterfullerene, C60): Spherical or ellipsoidal molecules composed entirely of carbon atoms, forming closed cages. The unique bonding configuration provides unique electronic and optical properties.
-
Carbon nanotubes: Cylindrical structures composed of rolled-up sheets of graphene (a single layer of graphite). Their exceptional strength-to-weight ratio and electrical conductivity make them promising materials for various applications.
These examples showcase how the fundamental characteristics arising from carbon's four valence electrons result in materials with drastically different properties and applications.
Understanding Carbon's Valence Electrons through Electronic Configuration
To further solidify our understanding, let's consider carbon's electronic configuration. Carbon's atomic number is 6, meaning it has six protons and six electrons. Its electronic configuration is 1s²2s²2p². The 1s and 2s orbitals are filled, while the 2p orbital contains only two electrons. However, when forming bonds, carbon promotes one electron from the 2s orbital to an empty 2p orbital, resulting in four unpaired electrons in the outermost shell (one in the 2s and three in the 2p orbitals). These four unpaired electrons are the valence electrons actively involved in chemical bonding. This hybridization of orbitals (sp³ hybridization in many cases) allows for the tetrahedral bonding geometry discussed earlier.
Applications and Future Directions
The implications of carbon's four valence electrons are profound and far-reaching. The field of materials science continues to explore novel carbon-based materials with exceptional properties. The development of new carbon-based polymers, composites, and nanomaterials holds immense promise for applications in energy storage, electronics, medicine, and many other fields.
Research into carbon's behavior under extreme conditions (high pressure, high temperature) continues to reveal new allotropes and functionalities. Understanding how carbon interacts with other elements and how its bonding can be manipulated is essential to designing materials with specific, tailored properties.
Conclusion
Carbon's four valence electrons are not just a numerical detail; they are the foundation of a vast and intricate world of chemistry and materials science. From the simplest hydrocarbons to the complex molecules of life and the innovative materials driving technological advancement, the remarkable versatility of carbon stems directly from its ability to form four covalent bonds. Further investigation into the intricacies of carbon's bonding and its ability to create diverse structures will undoubtedly continue to unveil new possibilities and shape the future of science and technology. Understanding this fundamental aspect of carbon's nature is crucial for anyone seeking to comprehend the fundamental building blocks of our universe and the possibilities they hold.
Latest Posts
Latest Posts
-
What Is A Reactant In Photosynthesis
Apr 03, 2025
-
32 Of 40 Percent Is What Number
Apr 03, 2025
-
What Is Found In Plant Cells But Not Animal Cells
Apr 03, 2025
-
What Percent Of 9 2 Is 43 7
Apr 03, 2025
-
What Is The Square Root Of 240
Apr 03, 2025
Related Post
Thank you for visiting our website which covers about Number Of Valence Electrons For Carbon . We hope the information provided has been useful to you. Feel free to contact us if you have any questions or need further assistance. See you next time and don't miss to bookmark.
