Most Reactive Group On The Periodic Table
listenit
Apr 02, 2025 · 5 min read
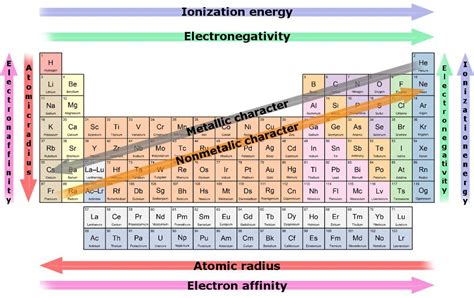
Table of Contents
The Most Reactive Group on the Periodic Table: Unveiling the Alkali Metals
The periodic table, a cornerstone of chemistry, organizes elements based on their atomic structure and properties. Understanding reactivity is crucial to comprehending the behavior of elements and their interactions. While several groups exhibit high reactivity, the alkali metals (Group 1) stand out as the most reactive group on the periodic table. This article delves deep into the reasons behind their exceptional reactivity, exploring their electronic configurations, reactions with various substances, and practical applications. We will also briefly compare their reactivity to other highly reactive groups to solidify our understanding.
The Electronic Configuration: The Key to Reactivity
The exceptional reactivity of alkali metals stems directly from their electronic configuration. Each alkali metal has a single electron in its outermost energy level, also known as the valence shell. This single valence electron is loosely held, making it relatively easy to remove. This is in stark contrast to other elements, many of which have multiple valence electrons or a complete outermost shell, leading to greater stability and lower reactivity.
Why a Single Electron Matters
This lone valence electron is crucial because atoms strive to achieve a stable electron configuration, typically resembling that of a noble gas (Group 18). Noble gases have a full outermost electron shell, making them exceptionally unreactive. Alkali metals, to achieve this noble gas configuration, readily lose their single valence electron, forming a +1 ion. This electron loss is energetically favorable, driving their high reactivity.
The ease with which they lose this electron is directly related to their position in the periodic table. As you move down Group 1, the atomic radius increases. This means the valence electron is further from the positively charged nucleus, experiencing weaker electrostatic attraction. Consequently, reactivity increases as you descend the group, with francium being the most reactive alkali metal.
Reactions with Various Substances: A Showcase of Reactivity
Alkali metals readily react with a wide range of substances, showcasing their extraordinary reactivity. Let's explore some key reactions:
1. Reaction with Water: A Vigorous Affair
The reaction of alkali metals with water is perhaps the most dramatic demonstration of their reactivity. When an alkali metal is placed in water, it reacts vigorously, producing hydrogen gas and a metal hydroxide. The reaction becomes increasingly vigorous as you move down the group.
- Lithium (Li): Reacts steadily, producing a gentle fizzing.
- Sodium (Na): Reacts more rapidly, producing a noticeable fizzing and often melting into a ball due to the heat generated.
- Potassium (K): Reacts violently, producing rapid fizzing, heat, and often igniting the hydrogen gas.
- Rubidium (Rb) and Cesium (Cs): React explosively with water, generating significant heat and potentially igniting the hydrogen gas.
The general equation for this reaction is:
2M(s) + 2H₂O(l) → 2MOH(aq) + H₂(g)
where M represents the alkali metal.
2. Reaction with Oxygen: Forming Oxides and Peroxides
Alkali metals also react readily with oxygen in the air. The nature of the product depends on the alkali metal and the reaction conditions. Lithium forms primarily lithium oxide (Li₂O), while sodium forms primarily sodium peroxide (Na₂O₂). Potassium, rubidium, and cesium form superoxides (e.g., KO₂).
3. Reaction with Halogens: Salt Formation
Alkali metals react vigorously with halogens (Group 17 elements) to form ionic salts. These reactions are highly exothermic, releasing significant amounts of heat. For example, the reaction of sodium with chlorine produces sodium chloride (common table salt):
2Na(s) + Cl₂(g) → 2NaCl(s)
4. Reaction with Acids: Highly Exothermic Reactions
The reaction of alkali metals with acids is highly exothermic, often leading to vigorous hydrogen gas evolution. The reaction is even more violent than the reaction with water. Safety precautions are paramount when conducting such experiments.
Comparing Alkali Metals to Other Highly Reactive Groups
While alkali metals are the most reactive group, it's important to compare their reactivity to other groups exhibiting high reactivity, such as the alkaline earth metals (Group 2) and the halogens (Group 17).
Alkali Metals vs. Alkaline Earth Metals
Alkaline earth metals have two valence electrons, making them less reactive than alkali metals. While they also react with water and oxygen, their reactions are generally less vigorous. The removal of two electrons requires more energy than the removal of a single electron.
Alkali Metals vs. Halogens
Halogens, despite their high reactivity, differ significantly in their reactivity mechanism. Halogens readily gain an electron to achieve a stable noble gas configuration, whereas alkali metals lose an electron. Halogens are strong oxidizing agents, readily accepting electrons from other elements, while alkali metals are strong reducing agents, readily donating electrons.
Practical Applications: Harnessing Reactivity
The high reactivity of alkali metals has led to various practical applications, despite the need for careful handling due to their inherent dangers. Some key applications include:
- Sodium in sodium-vapor lamps: Sodium's reactivity contributes to the bright yellow light emitted by these lamps.
- Lithium in batteries: Lithium's high electrochemical potential makes it ideal for use in lithium-ion batteries, powering many portable electronic devices.
- Potassium in fertilizers: Potassium compounds are essential nutrients for plant growth, making them crucial components of fertilizers.
- Cesium in atomic clocks: Cesium's precise atomic transitions are utilized in atomic clocks, providing highly accurate timekeeping.
Safety Precautions: Handling with Care
The high reactivity of alkali metals necessitates strict safety precautions during handling and storage. These metals should be handled under inert atmospheres (e.g., argon) to prevent reactions with air and moisture. Direct contact with skin or eyes should be avoided, as severe burns can occur. Appropriate personal protective equipment (PPE), including gloves, goggles, and lab coats, is essential when working with alkali metals. Proper waste disposal procedures must also be followed.
Conclusion: Understanding Reactivity's Significance
The alkali metals represent the most reactive group on the periodic table. Their high reactivity, stemming from their electronic configuration and the ease with which they lose their single valence electron, underpins their chemical behavior and diverse applications. Understanding their reactivity is not only crucial for understanding fundamental chemical principles but also for safe handling and harnessing their unique properties in various technological applications. Further research continues to explore new applications and improve our understanding of these fascinating elements. Remember always to prioritize safety when dealing with these highly reactive substances. Their remarkable properties come with significant hazards that must be carefully managed. The careful study and responsible application of the alkali metals will continue to shape technological advancements for years to come.
Latest Posts
Latest Posts
-
How Many Ounces Is In 3 4
Apr 03, 2025
-
What Is The Least Common Multiple Of 20 And 30
Apr 03, 2025
-
What Is The Square Root Of 170
Apr 03, 2025
-
How Long Does A Sensory Memory Last
Apr 03, 2025
-
63 Is 90 Percent Of What
Apr 03, 2025
Related Post
Thank you for visiting our website which covers about Most Reactive Group On The Periodic Table . We hope the information provided has been useful to you. Feel free to contact us if you have any questions or need further assistance. See you next time and don't miss to bookmark.
