Is Water Evaporating A Physical Change
listenit
Apr 02, 2025 · 6 min read
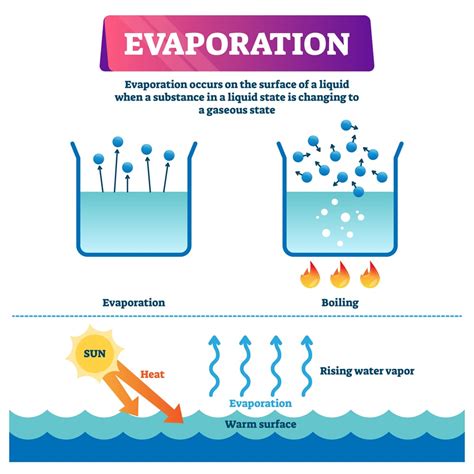
Table of Contents
Is Water Evaporating a Physical Change? A Deep Dive into the Science
Water evaporation is a ubiquitous process, shaping our weather, influencing our environment, and even playing a crucial role in our bodies. But is it a physical change or a chemical change? The answer, unequivocally, is physical. This article will delve deep into the science behind water evaporation, explaining why it's classified as a physical change and exploring the related concepts of boiling, condensation, and the role of energy in these phase transitions.
Understanding Physical and Chemical Changes
Before we dive into the specifics of water evaporation, let's establish a clear understanding of what differentiates physical and chemical changes.
Physical changes alter the form or appearance of a substance but don't change its chemical composition. Think about cutting paper, melting ice, or dissolving sugar in water. The substance remains the same; it's just rearranged or altered in its physical state. Crucially, these changes are often reversible. You can refreeze melted ice, and you can (with some effort) reassemble cut paper.
Chemical changes, on the other hand, involve a fundamental alteration in the chemical composition of a substance. New substances with different properties are formed. Burning wood, rusting iron, and cooking an egg are all examples of chemical changes. These changes are typically irreversible. You can't easily turn ash back into wood.
The Process of Water Evaporation: A Molecular Perspective
Water, in its liquid state, consists of molecules (H₂O) held together by relatively weak intermolecular forces. These forces are responsible for the cohesive properties of water, such as surface tension. However, these forces aren't strong enough to keep all the molecules together indefinitely.
Evaporation occurs when water molecules at the surface gain sufficient kinetic energy to overcome these intermolecular forces and escape into the gaseous phase (water vapor). This kinetic energy is typically acquired through heat absorption from the surrounding environment. The warmer the environment, the more kinetic energy the water molecules possess, and the faster the rate of evaporation.
Key aspects of evaporation:
- Surface Phenomenon: Evaporation primarily occurs at the surface of the liquid. The molecules deep within the liquid are surrounded by other molecules and experience strong intermolecular forces, making it harder for them to escape.
- Temperature Dependence: Higher temperatures lead to faster evaporation rates because the molecules have more kinetic energy.
- Surface Area: A larger surface area exposes more water molecules to the atmosphere, increasing the rate of evaporation.
- Air Movement: Wind or air currents remove water vapor molecules from the surface of the liquid, reducing the concentration of water vapor above the surface and allowing more molecules to escape. This accelerates evaporation.
- Humidity: High humidity (high concentration of water vapor in the air) slows down evaporation because the air is already saturated with water vapor.
Why Evaporation is a Physical Change
The key reason evaporation is considered a physical change is that the chemical composition of water remains unchanged. The water molecules in the gaseous state (water vapor) are the same H₂O molecules as those in the liquid state. No new chemical bonds are formed or broken during evaporation. The only change is the state of matter—from liquid to gas.
This is in stark contrast to a chemical change, where the molecules themselves are rearranged and transformed into different molecules with different properties. For example, if you electrolyze water, you break down H₂O molecules into hydrogen (H₂) and oxygen (O₂), representing a clear chemical change.
Boiling vs. Evaporation: Subtle Differences
While both boiling and evaporation involve the transition of water from liquid to gas, there are key distinctions:
- Temperature: Boiling occurs at a specific temperature (100°C or 212°F at standard atmospheric pressure), while evaporation can happen at any temperature below the boiling point.
- Location: Boiling involves the formation of vapor bubbles throughout the liquid, whereas evaporation primarily occurs at the surface.
- Rate: Boiling is generally a much faster process than evaporation.
Although boiling and evaporation differ in their mechanisms, both are physical changes because they involve only a change of state, not a change in the chemical composition of water.
Condensation: The Reverse of Evaporation
Condensation is the opposite of evaporation. It's the process by which water vapor transforms back into liquid water. This happens when water vapor molecules lose kinetic energy and the intermolecular forces become strong enough to hold them together in a liquid state.
Condensation is also a physical change because the chemical composition of water remains the same; it's merely a change in the state of matter from gas to liquid. The process is often observed on cool surfaces when humid air comes into contact with them.
The Role of Energy in Phase Transitions
Phase transitions, like evaporation and condensation, involve energy changes. Evaporation is an endothermic process, meaning it absorbs heat from the surroundings. The energy is used to overcome the intermolecular forces holding the water molecules together. Conversely, condensation is an exothermic process, releasing heat to the surroundings as the molecules come together and form liquid water.
The energy involved in these phase transitions is a crucial aspect of many natural processes, such as the water cycle, climate regulation, and even the cooling effect of sweating in animals.
Practical Applications and Real-World Examples
Understanding water evaporation as a physical change has significant practical applications in various fields:
- Weather Forecasting: Evaporation and condensation are fundamental components of the water cycle and are crucial in understanding weather patterns and predicting precipitation.
- Climate Science: Evaporation plays a significant role in regulating global temperatures and influencing climate patterns.
- Agriculture: Evaporation rates affect irrigation needs and crop growth.
- Industrial Processes: Many industrial processes rely on evaporation for purification, concentration, and drying.
- Cooling Systems: Sweating, a form of evaporation, provides a natural cooling mechanism for the human body and is the basis for many cooling technologies.
Conclusion: Evaporation – A Fundamental Physical Change
In conclusion, water evaporation is definitively a physical change. The process involves a transition of water from its liquid state to its gaseous state without altering its chemical composition. The only change is the state of matter, which is reversible through condensation. Understanding this fundamental physical change is essential in various scientific disciplines and has profound implications for our understanding of weather, climate, and various industrial and biological processes. The processes of evaporation and condensation, along with the energy changes associated with them, are fundamental aspects of the natural world and crucial for maintaining life as we know it. This seemingly simple process is a cornerstone of many larger, more complex phenomena, highlighting the power of understanding fundamental scientific concepts.
Latest Posts
Latest Posts
-
What Is 9 Ounces In Cups
Apr 03, 2025
-
Explain Why Chemical Equations Have To Be Balanced
Apr 03, 2025
-
How Do You Write 10 As A Fraction
Apr 03, 2025
-
Why Are Ionic Substances Soluble In Water
Apr 03, 2025
-
How Many Ml In A Dropper
Apr 03, 2025
Related Post
Thank you for visiting our website which covers about Is Water Evaporating A Physical Change . We hope the information provided has been useful to you. Feel free to contact us if you have any questions or need further assistance. See you next time and don't miss to bookmark.
