Is Water A Pure Substance Or Mixture
listenit
Apr 02, 2025 · 5 min read
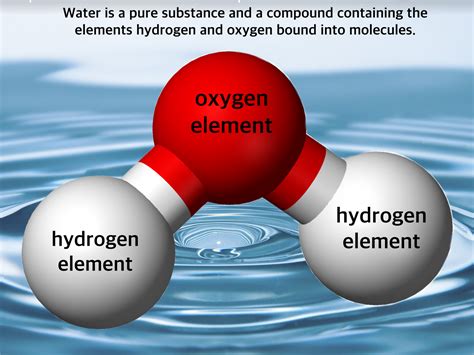
Table of Contents
Is Water a Pure Substance or a Mixture? A Deep Dive into the Chemistry of H₂O
The question, "Is water a pure substance or a mixture?" seems deceptively simple. After all, we readily associate water with purity, cleanliness, and life itself. However, the scientific answer requires a nuanced understanding of chemistry and the definitions of pure substances and mixtures. This article will delve into the complexities of water's composition, exploring its chemical nature and the factors that influence its purity. We'll examine different types of water, discuss the implications of its purity for various applications, and ultimately provide a definitive answer to the central question.
Understanding Pure Substances and Mixtures
Before we can classify water, we need to establish clear definitions of pure substances and mixtures.
Pure Substances: These are forms of matter with a uniform and unchanging composition. A pure substance cannot be separated into other substances by physical means, such as filtration or distillation. Pure substances can be further categorized into elements and compounds.
-
Elements: These are substances that cannot be broken down into simpler substances by chemical means. Examples include oxygen (O), hydrogen (H), and gold (Au).
-
Compounds: These are substances formed by the chemical combination of two or more elements in a fixed ratio. The properties of a compound are distinct from the properties of its constituent elements. Water (H₂O), for instance, is a compound formed by the chemical bonding of two hydrogen atoms and one oxygen atom.
Mixtures: Mixtures are combinations of two or more substances that are not chemically bonded. Unlike compounds, the components of a mixture retain their individual properties. Mixtures can be separated into their components by physical methods. Mixtures are further classified as homogeneous or heterogeneous.
-
Homogeneous Mixtures: These have a uniform composition throughout. For example, saltwater is a homogeneous mixture where salt is evenly distributed in water.
-
Heterogeneous Mixtures: These have a non-uniform composition, with different components visibly distinct. A mixture of sand and water is a heterogeneous mixture.
The Chemical Composition of Water
Water, with its chemical formula H₂O, is a compound, not a mixture. It is formed by the covalent bonding of two hydrogen atoms to a single oxygen atom. This bond is strong and requires chemical processes, not physical ones, to separate the hydrogen and oxygen. You can't simply filter out hydrogen from water; you need electrolysis or other chemical reactions.
This inherent chemical bonding signifies that water, in its purest form, is a pure substance. The ratio of hydrogen to oxygen is always 2:1. This fixed ratio distinguishes it from a mixture, where the ratio of components can vary.
Different Types of Water and their Purity
While pure water (H₂O) is a pure substance, the water we encounter in everyday life is rarely in its pure form. Several factors influence the purity of water:
1. Natural Water Sources: Water from rivers, lakes, oceans, and groundwater contains various dissolved substances, including minerals, salts, organic matter, and microorganisms. This makes natural water a mixture, not a pure substance. The composition of natural water varies widely depending on the source and geological factors.
2. Tap Water: Tap water undergoes treatment to remove impurities and improve its potability. However, it still contains small amounts of minerals and other substances, making it technically a mixture. The level of impurities varies depending on the water treatment process and the source water.
3. Distilled Water: This is water that has been purified through distillation, a process that involves boiling water and collecting the condensed steam. Distillation effectively removes most impurities, making distilled water much closer to pure H₂O than natural or tap water. However, it's still not perfectly pure, as trace impurities might remain.
4. Deionized Water: Deionized water is water that has had its ions (electrically charged atoms or molecules) removed. This is usually achieved using ion-exchange resins. Deionized water is extremely pure and is often used in scientific laboratories and industrial processes where high purity is crucial.
The Implications of Water Purity
The purity of water significantly impacts its applications:
-
Drinking Water: While some minerals in water are beneficial, excessive levels of impurities can pose health risks. Water treatment aims to ensure water is safe for consumption.
-
Industrial Processes: Many industrial processes require water of varying purity levels. High-purity water is essential in pharmaceutical manufacturing, semiconductor production, and power generation.
-
Scientific Research: Scientists use highly purified water in experiments and analyses to avoid interference from impurities.
-
Environmental Concerns: Water pollution from various sources introduces impurities into water bodies, harming aquatic life and potentially impacting human health. Maintaining water purity is critical for environmental sustainability.
Conclusion: Water – A Pure Substance at its Core
While the water we encounter daily often contains dissolved substances and is therefore a mixture, pure water (H₂O) is a pure substance, a compound formed from the chemical bonding of hydrogen and oxygen. The purity of water is relative and depends on its source and the degree of purification it has undergone. Understanding this distinction is crucial in various scientific, industrial, and environmental contexts. The quest for pure water highlights the significance of understanding chemical composition and its impact on the properties and applications of this essential substance. The simple molecule H₂O, while seemingly straightforward, holds complexities that continue to fascinate and challenge scientists and engineers alike. From the vast oceans to the tiniest cells, the ubiquitous nature of water, and its varying degrees of purity, underscore its central role in the world around us.
Latest Posts
Latest Posts
-
What Is The Shape Of The Orbits Of The Planets
Apr 03, 2025
-
1 1 2 Tablespoons Divided By 2
Apr 03, 2025
-
Lowest Common Multiple Of 25 And 35
Apr 03, 2025
-
Is Square Root Of 16 A Rational Number
Apr 03, 2025
-
What Is 6 8 As A Decimal
Apr 03, 2025
Related Post
Thank you for visiting our website which covers about Is Water A Pure Substance Or Mixture . We hope the information provided has been useful to you. Feel free to contact us if you have any questions or need further assistance. See you next time and don't miss to bookmark.
