Is Salt A Compound Element Or Mixture
listenit
Apr 05, 2025 · 5 min read
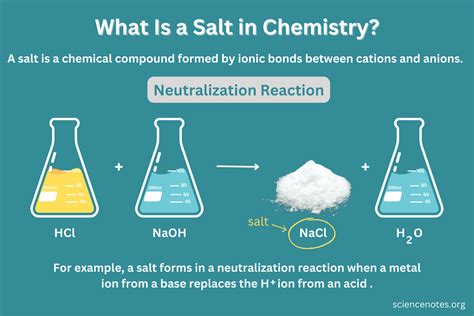
Table of Contents
Is Salt a Compound, Element, or Mixture? Understanding Chemical Composition
Salt, a ubiquitous substance in our kitchens and beyond, often sparks curiosity about its fundamental nature. Is it an element, a compound, or a mixture? Understanding this requires delving into the basic principles of chemistry and exploring the unique properties of sodium chloride, the common salt we use daily. This comprehensive guide will dissect the chemical makeup of salt, clarifying its classification and exploring related concepts.
What are Elements, Compounds, and Mixtures?
Before classifying salt, let's define the core terms:
Elements: The Building Blocks of Matter
Elements are fundamental substances that cannot be broken down into simpler substances by chemical means. They are composed of only one type of atom, characterized by its unique atomic number (the number of protons in its nucleus). Examples include oxygen (O), hydrogen (H), iron (Fe), and gold (Au). The periodic table organizes all known elements.
Compounds: Elements Bonding Together
Compounds are formed when two or more different elements chemically combine in fixed proportions. This combination involves the sharing or transfer of electrons between atoms, creating strong chemical bonds. The properties of a compound are distinctly different from the properties of its constituent elements. For example, water (H₂O) is a compound composed of hydrogen and oxygen, yet its properties are vastly different from those of hydrogen gas and oxygen gas.
Mixtures: A Blend of Substances
Mixtures are physical combinations of two or more substances, where each substance retains its individual chemical properties. Unlike compounds, mixtures do not have a fixed composition; the proportions of the components can vary. Mixtures can be homogeneous (uniform throughout, like saltwater) or heterogeneous (non-uniform, like sand and water). The components of a mixture can be separated by physical methods, such as filtration or distillation.
The Chemical Composition of Salt (Sodium Chloride)
Common table salt, also known as sodium chloride (NaCl), is a compound, not an element or a mixture. It's formed by the chemical bonding of two elements:
- Sodium (Na): A highly reactive alkali metal. In its pure elemental form, sodium is a soft, silvery-white metal that readily reacts with air and water.
- Chlorine (Cl): A highly reactive nonmetal gas, pale green in color, and toxic in its pure form.
These two elements, individually dangerous, combine to form sodium chloride, a stable and essential compound for life.
Ionic Bonding in NaCl
The bond between sodium and chlorine is an ionic bond. Sodium readily loses one electron to achieve a stable electron configuration, becoming a positively charged sodium ion (Na⁺). Chlorine readily gains one electron to also achieve a stable electron configuration, becoming a negatively charged chloride ion (Cl⁻). The electrostatic attraction between these oppositely charged ions forms the ionic bond that holds the sodium chloride crystal lattice together.
This crystal lattice structure is responsible for many of salt's physical properties, including its crystalline form, its high melting point, and its solubility in water.
Why Salt is Not an Element or a Mixture
Let's address why salt definitively isn't an element or a mixture:
-
Not an Element: Salt contains two different types of atoms (sodium and chlorine), making it impossible to be classified as a single element. Elements are defined by their unique atomic structure, containing only one type of atom.
-
Not a Mixture: The sodium and chlorine atoms in salt are chemically bonded, not simply physically mixed together. In a mixture, components can be separated by physical means, such as evaporation or filtration. Separating sodium and chlorine from sodium chloride requires a chemical process, such as electrolysis, demonstrating a chemical bond, not a physical mix. The properties of salt are distinctly different from the properties of its constituent elements, further confirming its compound nature.
The Importance of Salt in Biology and Industry
Sodium chloride's significance extends far beyond its culinary uses. It plays vital roles in:
Biological Roles:
- Electrolyte Balance: Salt is crucial for maintaining electrolyte balance in the human body, essential for nerve function, muscle contraction, and fluid regulation.
- Nutrient Absorption: Sodium ions participate in the absorption of nutrients in the digestive system.
- Osmosis and Cell Function: The concentration of sodium ions influences osmotic pressure, affecting the movement of water into and out of cells.
- Blood Pressure Regulation: While excessive salt intake can be detrimental, a proper balance is essential for healthy blood pressure.
Industrial Applications:
- Food Preservation: Salt's ability to inhibit microbial growth makes it a long-standing food preservative.
- De-icing: Salt's ability to lower the freezing point of water is used to de-ice roads and pavements in winter.
- Chemical Industry: Sodium chloride serves as a raw material in the production of various chemicals, including sodium hydroxide (lye), chlorine gas, and sodium metal.
- Water Softening: Salt is used in water softening systems to regenerate ion exchange resins.
Understanding the Properties of Salt: A Deeper Dive
The properties of sodium chloride directly reflect its ionic bonding and crystalline structure:
- Crystalline Structure: Salt forms a cubic crystal lattice, where sodium and chloride ions are arranged in a regular, repeating pattern.
- High Melting Point: The strong electrostatic attraction between sodium and chloride ions requires a significant amount of energy to break the ionic bonds, resulting in a high melting point.
- Solubility in Water: Water molecules, being polar, effectively interact with the charged sodium and chloride ions, dissolving the salt crystal lattice.
- Electrical Conductivity (when dissolved or molten): When dissolved in water or molten, the freely moving sodium and chloride ions allow the solution or melt to conduct electricity. In its solid crystalline form, salt does not conduct electricity.
Conclusion: Salt as a Chemical Compound
In conclusion, salt (sodium chloride) is unequivocally a chemical compound, formed by the ionic bonding of sodium and chlorine. It's not an element because it's composed of two different types of atoms, and it's not a mixture because its components are chemically bonded, not merely physically mixed. Its unique properties, stemming from its ionic bonding and crystalline structure, highlight its importance in biology and various industrial applications. Understanding the distinction between elements, compounds, and mixtures is fundamental to appreciating the complexity and beauty of chemistry and the world around us. This fundamental knowledge provides the groundwork for further exploration into the fascinating realm of chemical bonding and the properties of matter. Through this detailed analysis, we've established a clear and concise understanding of salt's chemical classification, solidifying its position as a quintessential chemical compound.
Latest Posts
Latest Posts
-
Balanced Equation For Copper And Nitric Acid
Apr 06, 2025
-
Two Satellites Are In Circular Orbits
Apr 06, 2025
-
Three Structural Isomers Have The Formula C5h12
Apr 06, 2025
-
The Four Nitrogenous Bases Found In Dna Are
Apr 06, 2025
-
Why Is Water Liquid At Room Temp
Apr 06, 2025
Related Post
Thank you for visiting our website which covers about Is Salt A Compound Element Or Mixture . We hope the information provided has been useful to you. Feel free to contact us if you have any questions or need further assistance. See you next time and don't miss to bookmark.
