In A Solution The Solvent Is
listenit
Apr 03, 2025 · 6 min read
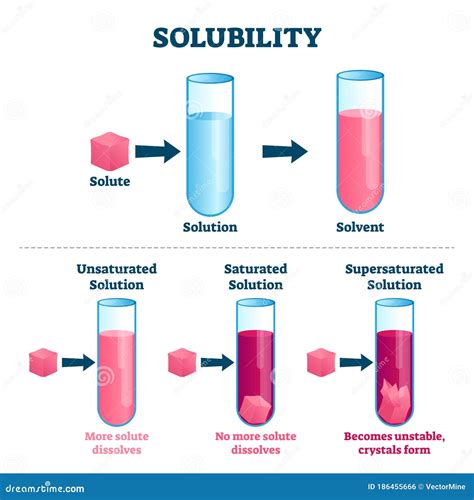
Table of Contents
In a Solution, the Solvent Is… Understanding the Fundamentals of Solutions
Understanding the fundamental components of a solution is crucial in various scientific disciplines, from chemistry and biology to environmental science and materials science. This comprehensive guide delves into the heart of solutions, focusing specifically on the role and characteristics of the solvent. We'll explore what a solvent is, its properties, the different types of solvents, and its critical importance in numerous applications.
What is a Solvent?
In simple terms, a solvent is the substance that dissolves a solute (another substance) to form a homogeneous mixture called a solution. Think of it as the "doing" part of the dissolving process. The solvent is typically present in a larger amount than the solute. The result of this interaction is a homogenous mixture where the solute is evenly distributed throughout the solvent, creating a single phase. This is in contrast to a heterogeneous mixture where distinct phases are visible.
A crucial aspect to understand is that the solvent's ability to dissolve a solute depends largely on the intermolecular forces between the solvent and solute molecules. "Like dissolves like" is a common adage used to describe this principle. Polar solvents, possessing a significant difference in electronegativity between atoms, tend to dissolve polar solutes. Conversely, nonpolar solvents, with a relatively even distribution of electron density, effectively dissolve nonpolar solutes. This principle guides our understanding of solubility and helps predict which solvents will be most effective for dissolving specific substances.
Properties of Solvents
Several key properties define the effectiveness and suitability of a solvent for a particular application. These properties include:
1. Polarity:
As mentioned earlier, polarity is a crucial property. Polar solvents have a positive and negative end, like water (H₂O), due to the unequal sharing of electrons. These solvents readily dissolve ionic compounds and polar molecules. Nonpolar solvents, like hexane (C₆H₁₄), lack this charge separation and are best at dissolving nonpolar molecules. The polarity of a solvent directly dictates its ability to interact with and dissolve different solutes.
2. Boiling Point:
The boiling point of a solvent is the temperature at which it transitions from a liquid to a gas. A higher boiling point indicates stronger intermolecular forces within the solvent. This property is essential when considering solvent recovery and purification. Solvents with lower boiling points are easier to evaporate and recover, making them advantageous in certain applications.
3. Viscosity:
Viscosity describes a solvent's resistance to flow. High-viscosity solvents flow slowly, while low-viscosity solvents flow easily. Viscosity influences the rate of dissolving and the ease of handling the solvent. Low viscosity is usually preferred for many applications.
4. Toxicity:
The toxicity of a solvent is a critical consideration, especially in biological and industrial settings. Some solvents are highly toxic and pose significant health risks, requiring careful handling and safety precautions. Choosing less toxic solvents is always preferable when alternatives exist.
5. Flammability:
The flammability of a solvent reflects its propensity to ignite and burn. Flammable solvents pose fire hazards and require careful storage and handling procedures. Using less flammable solvents minimizes the risk of fire-related incidents.
6. Miscibility:
Miscibility refers to the ability of two solvents to mix completely to form a homogeneous solution. Miscibility is crucial when dealing with mixtures of solvents. Understanding the miscibility of solvents is essential for predicting the behavior of solvent mixtures.
Types of Solvents
Solvents are categorized in various ways, based on their properties and applications. Some common classifications include:
1. Polar Protic Solvents:
These solvents possess a polar bond and a hydrogen atom bonded to an electronegative atom (like oxygen or nitrogen), allowing them to form hydrogen bonds. Water, methanol, and ethanol are examples of polar protic solvents. They are excellent for dissolving ionic compounds and polar molecules.
2. Polar Aprotic Solvents:
These solvents possess a polar bond but lack a hydrogen atom bonded to an electronegative atom, preventing hydrogen bond formation. Acetone, dimethyl sulfoxide (DMSO), and acetonitrile are examples. They are often used in organic reactions as they dissolve many organic compounds effectively.
3. Nonpolar Solvents:
These solvents have no significant dipole moment and do not form hydrogen bonds. Hexane, benzene, and toluene are examples. They are effective in dissolving nonpolar substances like fats and oils.
4. Organic Solvents:
These solvents are carbon-based and are widely used in organic chemistry. They encompass polar protic, polar aprotic, and nonpolar solvents. Examples include alcohols, ketones, ethers, and alkanes.
5. Inorganic Solvents:
These solvents are not carbon-based. Examples include water, liquid ammonia, and sulfuric acid. They are used in various applications, including inorganic chemistry and electrochemistry.
The Solvent's Role in Various Applications
The solvent plays a vital role in a multitude of applications:
1. Chemical Reactions:
Solvents are crucial in chemical reactions as they provide a medium for reactants to interact. The choice of solvent can significantly influence reaction rates, selectivity, and yield.
2. Pharmaceutical Industry:
Solvents are used extensively in the pharmaceutical industry for drug formulation, extraction, and purification. The solvent's properties must be carefully considered to ensure drug stability and safety.
3. Paint and Coatings:
Solvents are essential components of paints and coatings, providing the necessary viscosity and enabling the application and drying of the coating.
4. Cleaning and Extraction:
Solvents are used for cleaning and extraction processes in various industries, from cleaning equipment to extracting valuable compounds from natural sources.
5. Biological Systems:
Water, the universal solvent, is fundamental to all biological systems. It acts as a solvent for numerous biological molecules, facilitating metabolic processes and maintaining cellular integrity.
Choosing the Right Solvent: Key Considerations
Selecting the appropriate solvent requires careful consideration of several factors:
- Solubility of the solute: The solvent must effectively dissolve the target solute.
- Chemical compatibility: The solvent should not react with the solute or other components in the system.
- Safety: The solvent should have low toxicity and flammability.
- Cost: The solvent's cost should be economically viable for the application.
- Environmental impact: The solvent's environmental impact should be minimized.
- Boiling point and viscosity: These properties influence ease of handling and recovery.
Conclusion: The Unsung Hero of Solutions
The solvent, often overlooked, is the cornerstone of solutions. Its properties, such as polarity, boiling point, viscosity, and toxicity, dictate its suitability for a wide range of applications. Understanding the solvent's role is paramount in diverse fields, from chemistry and pharmaceuticals to environmental science and everyday life. Careful consideration of solvent properties is crucial for ensuring efficient, safe, and environmentally friendly processes. The careful selection of the appropriate solvent ensures the success of many scientific and industrial endeavors, highlighting its critical and often unsung role in our world. By understanding the nature and characteristics of solvents, we unlock a deeper comprehension of the intricacies of solutions and their widespread impact.
Latest Posts
Latest Posts
-
Common Factor Of 32 And 36
Apr 04, 2025
-
How To Find Mass Of A Planet
Apr 04, 2025
-
What Is Molar Volume Of Gas At Stp
Apr 04, 2025
-
Man Is The Only Animal That Blushes Or Needs To
Apr 04, 2025
-
In What Organelle Does Cellular Respiration Occur In
Apr 04, 2025
Related Post
Thank you for visiting our website which covers about In A Solution The Solvent Is . We hope the information provided has been useful to you. Feel free to contact us if you have any questions or need further assistance. See you next time and don't miss to bookmark.
