How To Calculate Mass Of Solute
listenit
Apr 02, 2025 · 6 min read
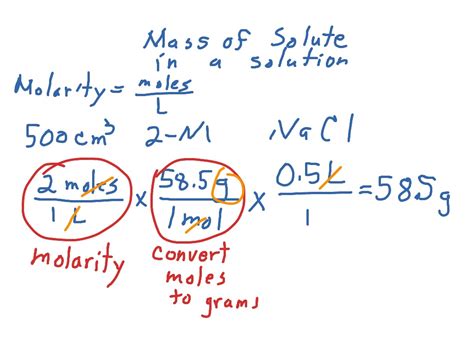
Table of Contents
How to Calculate the Mass of Solute: A Comprehensive Guide
Determining the mass of solute in a solution is a fundamental concept in chemistry with wide-ranging applications in various fields, from medicine and environmental science to industrial processes and culinary arts. Understanding how to calculate this crucial parameter is essential for accurately preparing solutions, interpreting experimental results, and solving various chemical problems. This comprehensive guide will walk you through various methods, providing clear explanations and practical examples to solidify your understanding.
Understanding Key Terms
Before delving into the calculations, let's define some key terms:
-
Solute: The substance that is dissolved in a solvent to form a solution. Think of it as the "stuff" that's being dissolved. Examples include salt (NaCl), sugar (sucrose), and various acids and bases.
-
Solvent: The substance that dissolves the solute. The most common solvent is water.
-
Solution: A homogeneous mixture of a solute and a solvent. This means the solute is evenly distributed throughout the solvent.
-
Mass: The amount of matter in a substance, typically measured in grams (g) or kilograms (kg).
-
Concentration: The amount of solute present in a given amount of solution. This is often expressed as molarity (moles of solute per liter of solution), molality (moles of solute per kilogram of solvent), or percentage by mass (mass of solute per mass of solution).
Methods for Calculating Mass of Solute
The method used to calculate the mass of the solute depends on the information you have available. Here are some common scenarios and their corresponding calculation methods:
1. Using Mass Percentage (%)
Mass percentage is a straightforward way to express concentration. It's defined as:
Mass Percentage (%) = (Mass of solute / Mass of solution) x 100%
To find the mass of the solute, rearrange the formula:
Mass of solute = (Mass Percentage / 100%) x Mass of solution
Example: A 10% (w/w) saline solution has a total mass of 500g. What is the mass of the solute (NaCl)?
Mass of solute = (10%/100%) x 500g = 50g
Therefore, the mass of NaCl in the solution is 50g.
2. Using Molarity (M)
Molarity is a widely used concentration unit, defined as the number of moles of solute per liter of solution:
Molarity (M) = Moles of solute / Liters of solution
To find the mass of the solute, you need to know its molar mass (grams per mole). The steps are as follows:
- Calculate moles of solute: Moles of solute = Molarity x Liters of solution
- Calculate mass of solute: Mass of solute = Moles of solute x Molar mass
Example: You have 2.0 L of a 0.5 M NaCl solution. The molar mass of NaCl is 58.44 g/mol. What is the mass of NaCl?
- Moles of NaCl = 0.5 M x 2.0 L = 1.0 mol
- Mass of NaCl = 1.0 mol x 58.44 g/mol = 58.44 g
Therefore, the mass of NaCl in the solution is 58.44 g.
3. Using Molality (m)
Molality is defined as the number of moles of solute per kilogram of solvent:
Molality (m) = Moles of solute / Kilograms of solvent
To find the mass of the solute, follow these steps:
- Calculate moles of solute: Moles of solute = Molality x Kilograms of solvent
- Calculate mass of solute: Mass of solute = Moles of solute x Molar mass
Example: You have a 1.0 m solution of glucose (C₆H₁₂O₆) in 500g (0.5kg) of water. The molar mass of glucose is 180.16 g/mol. What is the mass of glucose?
- Moles of glucose = 1.0 m x 0.5 kg = 0.5 mol
- Mass of glucose = 0.5 mol x 180.16 g/mol = 90.08 g
Therefore, the mass of glucose in the solution is 90.08 g.
4. Using Parts Per Million (ppm) or Parts Per Billion (ppb)
These units are used for extremely dilute solutions:
- ppm = (Mass of solute / Mass of solution) x 10⁶
- ppb = (Mass of solute / Mass of solution) x 10⁹
To find the mass of the solute, rearrange the formulas:
- Mass of solute = (ppm / 10⁶) x Mass of solution
- Mass of solute = (ppb / 10⁹) x Mass of solution
Example: A water sample has a lead concentration of 5 ppm and a total mass of 1000g. What is the mass of lead?
Mass of lead = (5 ppm / 10⁶) x 1000g = 0.005g or 5mg
5. Using Dilution Calculations
When diluting a concentrated solution, the amount of solute remains constant. This principle can be used to calculate the mass of solute in the diluted solution:
M₁V₁ = M₂V₂ (where M is molarity and V is volume)
This equation can be adapted to use mass instead of moles, provided you maintain consistent units throughout. For example:
(Mass of solute in concentrated solution / Volume of concentrated solution) = (Mass of solute in diluted solution / Volume of diluted solution)
Example: You have 100 mL of a 20% (w/v) solution. You dilute it to 500 mL. What is the mass of solute in the diluted solution?
Assuming 20% (w/v) means 20g solute per 100mL solution:
(20g / 100 mL) = (x g / 500 mL)
x = (20g x 500 mL) / 100 mL = 100g
Therefore, the diluted solution contains 100g of solute.
Advanced Scenarios and Considerations
The calculations described above are based on ideal conditions. In real-world situations, factors like temperature, solubility, and the presence of other substances can influence the accuracy of the results.
-
Solubility: The maximum amount of solute that can dissolve in a given amount of solvent at a specific temperature. If you attempt to dissolve more solute than the solvent can accommodate, the excess solute will remain undissolved.
-
Temperature: Temperature affects the solubility of most substances. Generally, increasing the temperature increases solubility.
-
Intermolecular forces: The interactions between solute and solvent molecules affect the solubility. "Like dissolves like" – polar solvents dissolve polar solutes, and nonpolar solvents dissolve nonpolar solutes.
Practical Applications
The ability to accurately calculate the mass of solute is crucial in various fields:
- Medicine: Preparing intravenous solutions, calculating drug dosages, and formulating pharmaceuticals.
- Environmental Science: Analyzing pollutant concentrations in water and soil samples.
- Food Science: Formulating food products, adjusting the concentration of ingredients, and controlling the properties of food.
- Industrial Chemistry: Controlling chemical reactions, optimizing industrial processes, and ensuring product quality.
Conclusion
Calculating the mass of solute is a fundamental skill in chemistry. This guide has provided a comprehensive overview of various methods and scenarios, empowering you to tackle a wide range of problems. Remember to always carefully consider the available information, choose the appropriate method, and pay attention to units to ensure accurate results. With practice and a solid understanding of the underlying principles, you can confidently master this essential skill and apply it to diverse scientific and practical contexts. Continue practicing with varied examples to deepen your understanding and proficiency. Remember to always double-check your work and use a calculator to avoid simple calculation errors.
Latest Posts
Latest Posts
-
1 1 2 Tablespoons Divided By 2
Apr 03, 2025
-
Lowest Common Multiple Of 25 And 35
Apr 03, 2025
-
Is Square Root Of 16 A Rational Number
Apr 03, 2025
-
What Is 6 8 As A Decimal
Apr 03, 2025
-
What Is 0 1 Repeating As A Fraction
Apr 03, 2025
Related Post
Thank you for visiting our website which covers about How To Calculate Mass Of Solute . We hope the information provided has been useful to you. Feel free to contact us if you have any questions or need further assistance. See you next time and don't miss to bookmark.
