How Monomers Are Related To Polymers
listenit
Apr 02, 2025 · 7 min read
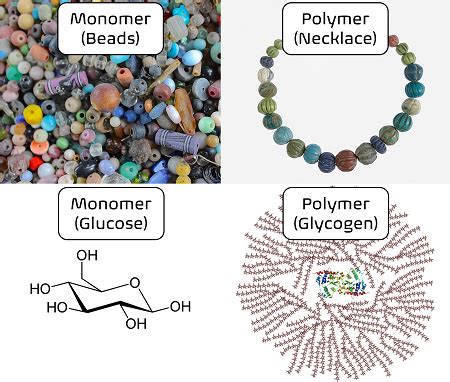
Table of Contents
How Monomers are Related to Polymers: A Deep Dive into Macromolecular Structure and Properties
The world around us is brimming with polymers. From the natural fibers in our clothing to the synthetic materials in our cars, polymers are ubiquitous. But what exactly are polymers, and how are they related to their building blocks, monomers? Understanding this fundamental relationship is key to appreciating the diverse properties and applications of these remarkable materials. This article explores the intricate connection between monomers and polymers, delving into their chemical structures, formation mechanisms, and the impact of monomer variations on polymer properties.
Understanding Monomers: The Building Blocks of Polymers
Monomers are small, relatively simple molecules that serve as the fundamental units for constructing larger, more complex molecules called polymers. Think of them as the individual Lego bricks that can be assembled to create elaborate structures. These monomers possess reactive functional groups – specific atoms or groups of atoms that participate in chemical reactions – allowing them to link together. The nature of these functional groups dictates how the monomers will bond and the characteristics of the resulting polymer. Common functional groups include:
-
Alkenes: Containing a carbon-carbon double bond (C=C), these are highly reactive and readily participate in addition polymerization. Examples include ethylene (used to make polyethylene) and propylene (used to make polypropylene).
-
Alcohols: Possessing a hydroxyl group (-OH), these can undergo condensation polymerization, forming ester or ether linkages. Examples include glucose (a monomer of cellulose) and lactic acid (a monomer of polylactic acid).
-
Amines: Containing an amino group (-NH2), these also participate in condensation polymerization, often reacting with carboxylic acids to form amide linkages (as in the formation of nylon).
-
Carboxylic Acids: Containing a carboxyl group (-COOH), these react with amines or alcohols in condensation polymerization.
Diverse Monomer Structures: A Wide Range of Possibilities
Monomers aren't just limited to a few simple structures. Their diversity stems from the vast number of possible arrangements of atoms and functional groups. This variation leads to a breathtaking array of polymer properties and functionalities. Consider the following examples:
-
Variations in Carbon Chain Length: The length of the carbon chain within a monomer significantly influences the resulting polymer's flexibility and strength. Longer chains often lead to more flexible polymers.
-
Branching: The presence of side branches on the monomer's carbon chain can significantly impact the polymer's density, crystallinity, and mechanical properties. Branched polymers are generally less dense and more flexible than linear polymers.
-
Functional Group Variations: Altering the type and position of functional groups on the monomer drastically changes the polymer's reactivity, solubility, and interactions with other molecules. This allows for the fine-tuning of polymer properties for specific applications.
-
Stereochemistry: The spatial arrangement of atoms around a chiral center in a monomer can lead to different polymer structures (isotactic, syndiotactic, atactic) with varying properties. For example, the stereochemistry of propylene monomers influences the properties of polypropylene, impacting its flexibility and crystallinity.
Polymerization: The Process of Monomer Assembly
Polymerization is the chemical process by which monomers link together to form long chains. There are two main types:
1. Addition Polymerization: Chain Growth
This mechanism involves monomers adding to a growing polymer chain without the loss of any atoms. It typically occurs with monomers containing double or triple bonds, such as alkenes. The process is initiated by a free radical, a cation, or an anion, which opens the double bond, creating a reactive site that attracts another monomer. This process repeats, resulting in a long chain molecule. Examples of addition polymers include:
- Polyethylene (PE): From ethylene monomers. Used in plastic bags, films, and bottles.
- Polypropylene (PP): From propylene monomers. Used in packaging, fibers, and containers.
- Polyvinyl Chloride (PVC): From vinyl chloride monomers. Used in pipes, flooring, and window frames.
- Polystyrene (PS): From styrene monomers. Used in packaging, insulation, and disposable cups.
- Polytetrafluoroethylene (PTFE) or Teflon: From tetrafluoroethylene monomers. Known for its non-stick properties, used in cookware and various industrial applications.
2. Condensation Polymerization: Step Growth
In contrast to addition polymerization, condensation polymerization involves the formation of a polymer chain with the elimination of a small molecule, such as water or methanol. This type of polymerization typically occurs with monomers containing two functional groups capable of reacting with each other, such as diacids and diamines. The reaction proceeds step-wise, with monomers reacting to form dimers, then trimers, and so on, until a long polymer chain is formed. Examples of condensation polymers include:
- Polyesters: Formed from the reaction of dicarboxylic acids and diols. Used in clothing fibers, plastic bottles, and films.
- Polyamides (Nylons): Formed from the reaction of diamines and diacids. Used in clothing fibers, carpets, and industrial applications.
- Polycarbonates: Formed from the reaction of phosgene and bisphenol A. Used in eyeglass lenses, compact discs, and automotive parts.
- Polyurethanes: Formed from the reaction of diisocyanates and polyols. Used in foams, coatings, and elastomers.
The Influence of Monomer Structure on Polymer Properties
The relationship between monomer structure and polymer properties is profound and multifaceted. Even subtle changes in the monomer can dramatically alter the resulting polymer's characteristics. These properties include:
-
Mechanical Strength: Linear polymers generally exhibit higher tensile strength than branched polymers due to their ability to form strong intermolecular interactions.
-
Flexibility and Elasticity: The presence of flexible segments in the polymer chain, such as long alkyl chains or ether linkages, contributes to increased flexibility and elasticity.
-
Melting Point and Glass Transition Temperature: These properties are influenced by the strength of intermolecular forces between polymer chains. Stronger forces lead to higher melting points and glass transition temperatures.
-
Solubility: The polarity of the monomer units affects the polymer's solubility. Polar monomers typically result in polar polymers that dissolve in polar solvents, while nonpolar monomers yield nonpolar polymers soluble in nonpolar solvents.
-
Crystallinity: The regularity of the polymer chain structure influences the degree of crystallinity. Regular, linear polymers tend to exhibit higher crystallinity than branched or irregular polymers. Crystalline polymers are typically stronger and more rigid than amorphous polymers.
Applications Based on Monomer-Polymer Relationships
The ability to control the properties of polymers by manipulating monomer structure is crucial for tailoring materials to specific applications. This precise control has revolutionized various industries, including:
-
Packaging: Polymers like polyethylene (PE), polypropylene (PP), and polystyrene (PS) are widely used in packaging due to their low cost, flexibility, and barrier properties.
-
Textiles: Polyesters, polyamides (nylons), and other synthetic fibers are used to create durable and comfortable clothing.
-
Construction: Polymers are used in construction materials such as pipes (PVC), insulation (polystyrene), and coatings.
-
Automotive: Polymers are utilized in various automotive components, such as dashboards, bumpers, and interior trim.
-
Biomedical: Biocompatible polymers are increasingly used in biomedical applications, including drug delivery systems, implants, and tissue engineering scaffolds. This often involves using monomers derived from natural sources.
Advanced Polymer Chemistry: Beyond Simple Linear Chains
The field of polymer chemistry extends far beyond simple linear chains. Advanced techniques allow for the synthesis of polymers with complex architectures, including:
-
Branched Polymers: These polymers exhibit side chains branching off the main chain, affecting properties like viscosity and solubility.
-
Cross-linked Polymers: These polymers contain covalent bonds between different polymer chains, enhancing their strength and rigidity. Vulcanized rubber is a classic example.
-
Block Copolymers: These consist of blocks of different monomers linked together, resulting in polymers with unique properties combining the characteristics of each block.
-
Graft Copolymers: These polymers have side chains of different monomers grafted onto a main chain.
Conclusion: A Continuous Evolution
The relationship between monomers and polymers is the cornerstone of polymer science and technology. The ability to design and synthesize polymers with specific properties by carefully selecting and manipulating monomers has unlocked countless applications across diverse sectors. As our understanding of this relationship deepens, we can anticipate even more innovative materials and technologies emerging from the remarkable world of polymers. Continued research into new monomers, polymerization techniques, and polymer architectures promises a future filled with even more sophisticated and versatile polymeric materials. The possibilities are truly limitless, constantly expanding the horizons of what's achievable with these versatile building blocks of the modern world.
Latest Posts
Latest Posts
-
What Is 9 Ounces In Cups
Apr 03, 2025
-
Explain Why Chemical Equations Have To Be Balanced
Apr 03, 2025
-
How Do You Write 10 As A Fraction
Apr 03, 2025
-
Why Are Ionic Substances Soluble In Water
Apr 03, 2025
-
How Many Ml In A Dropper
Apr 03, 2025
Related Post
Thank you for visiting our website which covers about How Monomers Are Related To Polymers . We hope the information provided has been useful to you. Feel free to contact us if you have any questions or need further assistance. See you next time and don't miss to bookmark.
