How Many Valence Electrons In Si
listenit
Mar 31, 2025 · 5 min read
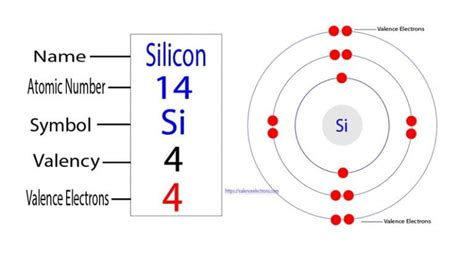
Table of Contents
How Many Valence Electrons Does Silicon (Si) Have? A Deep Dive into Electronic Structure
Silicon, a cornerstone element in modern technology, plays a crucial role in semiconductors, solar cells, and countless other applications. Understanding its electronic structure, particularly the number of valence electrons, is paramount to comprehending its behavior and properties. This comprehensive guide will delve deep into the question: How many valence electrons does silicon (Si) have? We'll explore the underlying principles of electron configuration, the significance of valence electrons, and the implications for silicon's unique characteristics.
Understanding Electron Configuration and Valence Electrons
Before we pinpoint the number of valence electrons in silicon, let's establish a fundamental understanding of electron configuration. Atoms consist of a nucleus containing protons and neutrons, surrounded by electrons orbiting in various energy levels or shells. These shells are organized into subshells (s, p, d, f), each capable of holding a specific number of electrons. The electron configuration represents the arrangement of electrons within these shells and subshells.
The valence electrons are the electrons located in the outermost shell of an atom. These electrons are the most loosely bound and, therefore, are the most involved in chemical bonding and reactions. The number of valence electrons significantly dictates an element's chemical properties and reactivity. Elements within the same group (vertical column) on the periodic table share the same number of valence electrons, resulting in similar chemical behavior.
Determining Silicon's Valence Electrons
Silicon (Si) has an atomic number of 14, meaning it possesses 14 protons and 14 electrons in a neutral atom. To determine its electron configuration, we follow the Aufbau principle, filling orbitals according to their energy levels:
- 1s²: The first shell (n=1) contains the 1s subshell, holding a maximum of 2 electrons.
- 2s²: The second shell (n=2) begins with the 2s subshell, also holding a maximum of 2 electrons.
- 2p⁶: The second shell also contains the 2p subshell, which can accommodate up to 6 electrons.
- 3s²: The third shell (n=3) starts with the 3s subshell, holding another 2 electrons.
- 3p²: Finally, the remaining 2 electrons occupy the 3p subshell.
Therefore, the complete electron configuration of silicon is 1s²2s²2p⁶3s²3p².
The outermost shell of silicon is the third shell (n=3), which contains both the 3s and 3p subshells. Adding the electrons in these subshells (2 + 2 = 4), we find that silicon has 4 valence electrons.
Visualizing Silicon's Valence Electrons
It's helpful to visualize silicon's electronic structure. Imagine the nucleus at the center, surrounded by three shells. The first two shells are completely filled, while the third shell has four electrons – these are the valence electrons actively participating in chemical bonding. This arrangement explains silicon's ability to form four covalent bonds, a crucial aspect of its semiconducting properties.
The Significance of Silicon's Four Valence Electrons
The presence of four valence electrons is the key to understanding silicon's unique properties and its widespread use in technology. Let's explore how this impacts its behavior:
1. Semiconducting Properties:
Silicon's four valence electrons allow it to form a tetrahedral structure in its crystalline form. Each silicon atom shares its four valence electrons with four neighboring silicon atoms, creating a strong covalent network. This structure is responsible for silicon's semiconducting behavior. At absolute zero temperature, silicon is an insulator; however, at higher temperatures, some electrons gain enough energy to jump into a higher energy level, creating electron-hole pairs and enabling electrical conductivity. This controlled conductivity is what makes silicon indispensable in transistors and integrated circuits.
2. Chemical Bonding:
The four valence electrons readily participate in covalent bonding with other atoms, including oxygen, hydrogen, and other silicon atoms. This ability to form strong covalent bonds contributes to the stability and durability of silicon-based materials. Silicon dioxide (SiO₂), for example, is a highly stable compound used in various applications, including insulation in integrated circuits.
3. Doping and Semiconductor Technology:
The controlled introduction of impurities (doping) into the silicon crystal lattice further manipulates its semiconducting properties. Doping with elements containing either three (like boron) or five (like phosphorus) valence electrons alters the conductivity of silicon, creating either p-type or n-type semiconductors, respectively. This controlled manipulation of conductivity is fundamental to the creation of transistors, diodes, and other electronic components.
4. Applications in Solar Cells:
Silicon's ability to absorb sunlight and convert it into electricity makes it a key material in solar cell technology. The interaction of photons with silicon's electrons generates electron-hole pairs, resulting in an electric current. The efficiency of silicon-based solar cells continues to improve, making solar energy a more viable and sustainable power source.
Comparing Silicon to Other Elements
Comparing silicon's valence electrons to other elements in the same group (Group 14) highlights the trends in chemical properties. Carbon, with four valence electrons, forms diverse organic compounds due to its ability to form long chains and rings. Germanium, also with four valence electrons, shares similar semiconducting properties to silicon but has a higher intrinsic conductivity. Tin and lead, although having four valence electrons, exhibit more metallic characteristics due to the increasing influence of inner electrons and weaker covalent bonding.
Conclusion: The Importance of Valence Electrons in Silicon
The answer to "How many valence electrons does silicon have?" is definitively four. This seemingly simple number dictates silicon's unique properties and its pivotal role in modern technology. Its semiconducting behavior, stemming from its four valence electrons and ability to form a stable tetrahedral structure, underpins the operation of countless electronic devices. From transistors to solar cells, silicon's impact on our daily lives is undeniable, highlighting the profound influence of valence electrons on an element's characteristics and applications. Further research continues to explore new and innovative ways to harness silicon's potential, pushing the boundaries of technological advancement. Understanding the fundamental role of its four valence electrons is the key to unlocking this potential.
Latest Posts
Latest Posts
-
What Is 3 20 As A Percentage
Apr 02, 2025
-
What Is 2 And 1 4 As An Improper Fraction
Apr 02, 2025
-
What Is 18 24 As A Percent
Apr 02, 2025
-
How Do You Graph X 6
Apr 02, 2025
-
2000 Mg Equals How Many G
Apr 02, 2025
Related Post
Thank you for visiting our website which covers about How Many Valence Electrons In Si . We hope the information provided has been useful to you. Feel free to contact us if you have any questions or need further assistance. See you next time and don't miss to bookmark.
