How Many Valence Electrons Does Cd Have
listenit
Apr 01, 2025 · 5 min read
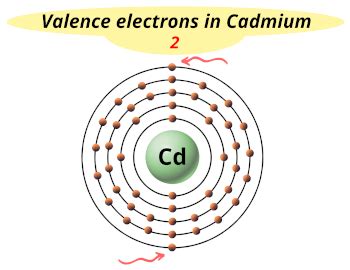
Table of Contents
How Many Valence Electrons Does Cd Have? Exploring the Electronic Structure of Cadmium
Cadmium (Cd), a transition metal with a rich history and diverse applications, holds a unique position in the periodic table. Understanding its electronic structure, particularly the number of valence electrons, is crucial to comprehending its chemical behavior and properties. This comprehensive guide delves deep into the electronic configuration of cadmium, explaining how to determine its valence electrons and how this impacts its reactivity and applications.
Understanding Valence Electrons
Before we dive into the specifics of cadmium, let's establish a foundational understanding of valence electrons. Valence electrons are the electrons located in the outermost shell of an atom. These electrons are responsible for the chemical bonding behavior of an element, determining its reactivity and the types of compounds it can form. They are the key players in chemical reactions, easily gained, lost, or shared with other atoms.
The number of valence electrons an atom possesses is directly related to its position in the periodic table. Elements within the same group (vertical column) have the same number of valence electrons, leading to similar chemical properties. This predictable pattern simplifies the process of determining valence electrons for many elements.
Determining the Valence Electrons of Cadmium (Cd)
Cadmium's atomic number is 48, meaning it has 48 protons and 48 electrons in a neutral atom. To determine the number of valence electrons, we need to examine its electronic configuration. This configuration describes how electrons are distributed among the different energy levels (shells) and subshells within the atom.
The electronic configuration of cadmium is [Kr] 4d<sup>10</sup> 5s<sup>2</sup>.
Let's break this down:
-
[Kr]: This represents the electron configuration of krypton (Kr), a noble gas. Noble gases have a complete outermost electron shell, making them exceptionally stable and unreactive. Using the noble gas core shorthand simplifies the representation of electron configurations.
-
4d<sup>10</sup>: This indicates that the 4d subshell is completely filled with 10 electrons. The 4d subshell is considered an inner shell in the context of valence electrons for cadmium.
-
5s<sup>2</sup>: This indicates that the 5s subshell contains 2 electrons. This is cadmium's outermost shell.
Therefore, cadmium has two valence electrons. These two electrons in the 5s subshell are the ones primarily involved in chemical bonding.
The Significance of Two Valence Electrons in Cadmium's Chemistry
The presence of only two valence electrons significantly influences cadmium's chemical behavior. Unlike elements with many valence electrons, cadmium exhibits relatively low reactivity. This is because it is more energetically favorable for cadmium to lose two electrons and achieve a stable, filled 4d subshell, rather than gaining six more to achieve a full 5p subshell.
This tendency to lose two electrons explains why cadmium typically forms compounds with an oxidation state of +2. This means it loses two electrons during chemical reactions, forming ions with a 2+ charge (Cd<sup>2+</sup>). These ions are involved in various chemical compounds and reactions.
Applications Leveraging Cadmium's Electronic Structure
Cadmium's unique electronic structure and consequent properties underpin its applications in various fields:
1. Batteries:
Cadmium's ability to readily accept and release electrons makes it a valuable component in rechargeable nickel-cadmium (NiCd) batteries. These batteries were widely used in portable electronics for decades, leveraging cadmium's electrochemical properties for efficient energy storage. However, concerns about cadmium's toxicity have led to its decreased use in favor of more environmentally friendly alternatives.
2. Pigments:
Cadmium compounds, particularly cadmium sulfide (CdS) and cadmium selenide (CdSe), are vibrant pigments used in paints, plastics, and other materials. The color of these pigments is directly linked to the electronic transitions within the cadmium-containing compounds, a phenomenon influenced by cadmium's electronic structure. However, similar to battery applications, environmental concerns related to cadmium's toxicity limit their use in certain applications.
3. Nuclear Reactors:
Cadmium's ability to absorb neutrons efficiently has led to its use in nuclear reactors as a control rod material. The absorption of neutrons is a nuclear process that helps regulate the rate of fission reactions, ensuring the safe and efficient operation of nuclear reactors. This application takes advantage of the specific interactions between neutrons and cadmium's atomic structure, which is partly governed by its electronic configuration.
Cadmium's Toxicity and Environmental Concerns
Despite its useful properties, cadmium is a highly toxic element. Exposure to cadmium can lead to various health problems, including kidney damage, bone disease, and lung cancer. This toxicity necessitates careful handling and disposal of cadmium-containing materials, and has driven efforts to find safer alternatives in many of its applications.
Comparison with Other Group 12 Elements: Zinc and Mercury
Cadmium belongs to Group 12 of the periodic table, along with zinc (Zn) and mercury (Hg). All three elements have two valence electrons, but their properties differ significantly due to variations in their electronic configurations and atomic sizes.
-
Zinc (Zn): Zinc also has two valence electrons ([Ar] 3d<sup>10</sup> 4s<sup>2</sup>) and shows a similar +2 oxidation state. However, zinc is generally less toxic than cadmium and finds wider applications.
-
Mercury (Hg): Mercury also has two valence electrons ([Xe] 4f<sup>14</sup> 5d<sup>10</sup> 6s<sup>2</sup>). However, unlike zinc and cadmium, mercury is a liquid at room temperature due to weak metallic bonding. It is highly toxic and poses significant environmental concerns.
Conclusion: Cadmium's Valence Electrons and Their Impact
In conclusion, cadmium possesses two valence electrons, a defining characteristic that significantly influences its chemical behavior, applications, and toxicity. Understanding its electronic structure provides insight into its reactivity, allowing for the development of specific applications while also highlighting the importance of safety measures and environmentally responsible practices when handling cadmium and its compounds. While its uses in certain technologies are being phased out due to its toxicity, the fundamental knowledge of its electronic configuration remains vital for advancements in various scientific fields and technological developments. The ongoing research into safer alternatives underlines the continuous efforts to harness the benefits of elements like cadmium while mitigating their potential risks.
Latest Posts
Latest Posts
-
Common Multiple Of 2 3 5
Apr 03, 2025
-
What Is 5 6 As A Decimal
Apr 03, 2025
-
The Conjugate Base Of Hso4 Is
Apr 03, 2025
-
Examples Of Gay Lussacs Law In Real Life
Apr 03, 2025
-
Law Of Sines And Law Of Cosines Word Problems
Apr 03, 2025
Related Post
Thank you for visiting our website which covers about How Many Valence Electrons Does Cd Have . We hope the information provided has been useful to you. Feel free to contact us if you have any questions or need further assistance. See you next time and don't miss to bookmark.
