How Many Valence Electrons Are In Cl
listenit
Apr 05, 2025 · 5 min read
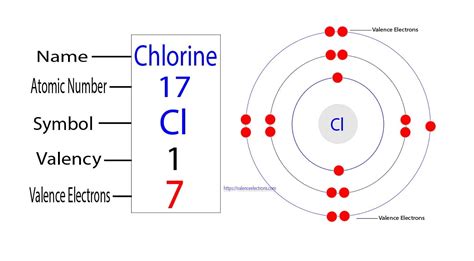
Table of Contents
How Many Valence Electrons Are in Cl? Understanding Chlorine's Reactivity
Chlorine (Cl), a ubiquitous element found in everyday life and crucial to various industrial processes, holds a fascinating position in the periodic table. Understanding its electronic structure, particularly its valence electrons, is key to comprehending its chemical behavior and reactivity. This article delves deep into the question: how many valence electrons are in Cl? and explores the implications of this number on chlorine's properties and its role in the world around us.
What are Valence Electrons?
Before we answer the central question, let's clarify what valence electrons are. Valence electrons are the electrons located in the outermost shell, or energy level, of an atom. These electrons are crucial because they determine an atom's reactivity and its ability to form chemical bonds with other atoms. They are the primary participants in chemical reactions, readily engaging in sharing or transferring with other atoms to achieve a stable electron configuration, often fulfilling the octet rule (eight electrons in the outermost shell).
Determining Chlorine's Valence Electrons
Chlorine's atomic number is 17, meaning it possesses 17 protons and 17 electrons in a neutral atom. To determine the number of valence electrons, we need to examine its electron configuration. The electron configuration of chlorine is 1s²2s²2p⁶3s²3p⁵.
This configuration tells us the distribution of electrons across different energy levels (shells):
- 1s²: Two electrons in the first energy level (shell).
- 2s²2p⁶: Eight electrons in the second energy level.
- 3s²3p⁵: Seven electrons in the third energy level.
The outermost shell of chlorine is the third energy level (n=3), containing seven electrons (3s²3p⁵). Therefore, chlorine has 7 valence electrons.
The Significance of Seven Valence Electrons
The presence of seven valence electrons significantly influences chlorine's chemical behavior. Atoms strive for stability, often by achieving a full outermost shell, resembling the electron configuration of a noble gas. For chlorine, achieving a stable octet (eight electrons) requires gaining one electron. This propensity to gain an electron makes chlorine highly reactive and explains its strong electronegativity.
Electronegativity and Reactivity
Electronegativity is a measure of an atom's ability to attract electrons towards itself in a chemical bond. Chlorine's high electronegativity stems directly from its seven valence electrons. It readily attracts electrons from other atoms to complete its octet, resulting in the formation of ionic or covalent bonds.
Ionic Bonding: Chloride Ion Formation
When chlorine reacts with a metal, such as sodium (Na), it readily accepts an electron from sodium, forming a chloride ion (Cl⁻). Sodium, with one valence electron, loses this electron to become a sodium ion (Na⁺). The electrostatic attraction between the positively charged sodium ion and the negatively charged chloride ion forms an ionic bond, resulting in the formation of sodium chloride (NaCl), commonly known as table salt.
Understanding the ionic bond formation between sodium and chlorine is crucial to understanding the role of valence electrons in chemical bonding. Sodium's one valence electron is readily transferred to chlorine's incomplete outermost shell, creating a stable ionic compound.
Covalent Bonding: Sharing Electrons
Chlorine can also form covalent bonds by sharing electrons with other non-metal atoms. In a covalent bond, atoms share electrons to achieve a stable electron configuration. For example, in a chlorine molecule (Cl₂), two chlorine atoms share one pair of electrons, allowing each atom to effectively have eight electrons in its outermost shell. This electron sharing completes the octet for both atoms and leads to a strong covalent bond between them.
Chlorine's Role in Various Applications
Chlorine's unique properties, directly linked to its seven valence electrons and high reactivity, make it vital in a myriad of applications:
Water Purification
Chlorine is extensively used in water purification to disinfect water supplies and eliminate harmful bacteria and viruses. Its strong oxidizing properties effectively kill microorganisms, ensuring safe drinking water for millions of people globally. This sterilization process relies on chlorine's reactivity to disrupt the cellular functions of these harmful organisms.
Industrial Processes
Chlorine plays a critical role in numerous industrial processes, including the production of plastics (PVC), solvents, and various chemicals. Its reactivity allows it to participate in a vast array of chemical reactions, facilitating the synthesis of essential materials for various industries.
Medical Applications
While chlorine gas is toxic, various chlorine-containing compounds find applications in medicine. These compounds often exploit chlorine's reactivity to achieve desired medicinal effects, illustrating the controlled use of this reactive element in beneficial ways.
Safety Considerations
It's crucial to emphasize that chlorine gas (Cl₂) is highly toxic and corrosive. Exposure to chlorine gas can cause severe respiratory problems and even death. Safe handling practices and appropriate protective measures are essential when working with chlorine or chlorine-containing compounds.
Conclusion: The Importance of Valence Electrons
The number of valence electrons in an atom is a fundamental property determining its chemical behavior and reactivity. Chlorine, with its seven valence electrons, showcases the significance of this concept. Its high electronegativity, ability to form both ionic and covalent bonds, and subsequent use in various applications all stem from this fundamental characteristic. Understanding the implications of chlorine's seven valence electrons is key to appreciating its wide-ranging roles in our world, from purifying drinking water to facilitating industrial processes. The reactivity and properties of chlorine are a direct consequence of its electronic structure, highlighting the importance of studying the valence electrons of elements to predict their behavior and harness their potential for beneficial applications.
The detailed exploration of chlorine's valence electrons provides a clear illustration of the fundamental principles of chemical bonding and reactivity. This knowledge forms the foundation for understanding more complex chemical systems and processes. Further studies into the periodic table and the electronic configurations of elements will enhance one's understanding of chemical behavior and the vast applications of chemistry in various fields. The simplicity of understanding the valence electrons allows for a gateway to more complex concepts in chemistry and opens a fascinating world of exploration into the structure and properties of matter.
Latest Posts
Latest Posts
-
Is Milk Turning Sour A Chemical Change
Apr 05, 2025
-
What Is The Low Point Of A Wave Called
Apr 05, 2025
-
What Is The Derivative Of Cos 2x
Apr 05, 2025
-
What Part Of An Atom Has A Positive Charge
Apr 05, 2025
-
How To Find Where The Tangent Line Is Horizontal
Apr 05, 2025
Related Post
Thank you for visiting our website which covers about How Many Valence Electrons Are In Cl . We hope the information provided has been useful to you. Feel free to contact us if you have any questions or need further assistance. See you next time and don't miss to bookmark.
