How Many Unpaired Electrons Are In Sulfur
listenit
Apr 02, 2025 · 5 min read
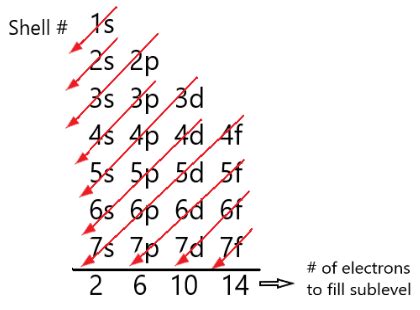
Table of Contents
How Many Unpaired Electrons Are in Sulfur? A Deep Dive into Atomic Structure and Electron Configuration
Sulfur, a vibrant yellow nonmetal crucial to life and numerous industrial processes, presents an intriguing case study in atomic structure. Understanding its electron configuration is key to comprehending its chemical behavior, reactivity, and unique properties. This article delves deep into the question: how many unpaired electrons are in sulfur? We'll explore the fundamental principles of electron configuration, delve into the specifics of sulfur's atomic structure, and examine the implications of its unpaired electrons.
Understanding Electron Configuration: The Foundation
Before we tackle sulfur specifically, let's establish a firm grasp of electron configuration. This principle dictates how electrons are arranged within an atom's electron shells and subshells. The arrangement is governed by several fundamental rules:
- Aufbau Principle: Electrons fill the lowest energy levels first. This is like building a house – you start with the foundation before adding the upper floors.
- Pauli Exclusion Principle: Each orbital can hold a maximum of two electrons, but they must have opposite spins (represented as ↑ and ↓). Think of it as two roommates sharing an apartment, but they need to have different personalities (spins).
- Hund's Rule: Within a subshell, electrons will occupy individual orbitals before pairing up in the same orbital. This maximizes electron spin, promoting stability. It's like students filling individual desks in a classroom before doubling up at the same desk.
These rules, applied systematically, allow us to predict the electron configuration of any element, including sulfur.
Sulfur's Atomic Structure and Electron Configuration
Sulfur (S) has an atomic number of 16, meaning it possesses 16 protons and 16 electrons in a neutral atom. To determine its electron configuration, we follow the Aufbau principle and fill the electron shells and subshells accordingly:
1s² 2s² 2p⁶ 3s² 3p⁴
Let's break this down:
- 1s²: The first shell (n=1) contains one subshell (s), which holds a maximum of two electrons.
- 2s²: The second shell (n=2) contains an s subshell holding two electrons.
- 2p⁶: The second shell also contains a p subshell, which can accommodate up to six electrons.
- 3s²: The third shell (n=3) begins with an s subshell holding two electrons.
- 3p⁴: The third shell's p subshell contains four electrons.
Identifying Unpaired Electrons in Sulfur
Now, to answer the central question, let's visualize the 3p subshell:
The 3p subshell has three orbitals (3px, 3py, 3pz), each capable of holding two electrons. According to Hund's rule, electrons will first occupy each orbital individually before pairing up. With four electrons in the 3p subshell, we have the following arrangement:
3px ↑ 3py ↑ 3pz ↑↓
Notice that two electrons are unpaired (one in 3px and one in 3py), while the remaining two are paired in the 3pz orbital.
Therefore, sulfur has two unpaired electrons.
Implications of Unpaired Electrons in Sulfur's Properties
The presence of unpaired electrons significantly influences sulfur's chemical and physical properties. These unpaired electrons are responsible for:
-
Paramagnetism: Sulfur is paramagnetic, meaning it is weakly attracted to a magnetic field. This is a direct consequence of the unpaired electrons' spins. Substances with all paired electrons are diamagnetic and are repelled by a magnetic field.
-
Reactivity: The unpaired electrons make sulfur readily reactive. It readily forms covalent bonds with other atoms to achieve a stable octet configuration, where all its valence electrons are paired. This explains its diverse range of compounds.
-
Variable Oxidation States: Sulfur exhibits several oxidation states, from -2 to +6. This ability to lose or gain electrons is directly linked to the presence of unpaired electrons, allowing for varied bonding arrangements.
-
Color: The presence of unpaired electrons often contributes to the color of substances. The characteristic yellow color of sulfur is partly due to electronic transitions involving these unpaired electrons.
Sulfur's Role in Biological and Industrial Processes
The unique properties of sulfur, stemming from its electronic configuration and unpaired electrons, render it essential in numerous biological and industrial processes.
Biological Significance:
-
Amino Acids: Sulfur is a crucial component of several amino acids, including cysteine and methionine, which are fundamental building blocks of proteins. The sulfur atoms in these amino acids participate in disulfide bonds, crucial for protein structure and function.
-
Enzymes: Many enzymes require sulfur for their catalytic activity. The sulfur atoms can act as electron donors or acceptors, facilitating biochemical reactions.
-
Vitamins: Biotin (Vitamin B7) contains sulfur and plays a vital role in metabolism.
Industrial Applications:
-
Sulfuric Acid Production: Sulfur is a fundamental raw material in the production of sulfuric acid, arguably the world's most important industrial chemical. Sulfuric acid is used extensively in fertilizer production, metal processing, and numerous other applications.
-
Vulcanization of Rubber: The addition of sulfur to rubber during vulcanization enhances its strength, durability, and elasticity, leading to the production of durable tires and other rubber products.
-
Pharmaceuticals: Sulfur compounds find applications in various pharmaceuticals, owing to their antimicrobial and other therapeutic properties.
-
Fertilizers: Sulfur is a vital nutrient for plant growth. Sulfate fertilizers are used to supplement sulfur levels in soils, supporting agricultural productivity.
Conclusion: The Significance of Unpaired Electrons in Sulfur
In conclusion, sulfur possesses two unpaired electrons, located in its 3p subshell. This seemingly small detail profoundly influences its chemical behavior, reactivity, and wide-ranging applications in biological and industrial processes. Understanding sulfur's electron configuration offers a crucial insight into its properties and underscores the importance of atomic structure in determining the characteristics of elements and their compounds. This knowledge is fundamental in various scientific disciplines, from chemistry and biochemistry to materials science and engineering. The unpaired electrons are not just a detail; they are the key to unlocking sulfur's multifaceted role in the world around us.
Latest Posts
Latest Posts
-
How Many Ounces Is In 3 4
Apr 03, 2025
-
What Is The Least Common Multiple Of 20 And 30
Apr 03, 2025
-
What Is The Square Root Of 170
Apr 03, 2025
-
How Long Does A Sensory Memory Last
Apr 03, 2025
-
63 Is 90 Percent Of What
Apr 03, 2025
Related Post
Thank you for visiting our website which covers about How Many Unpaired Electrons Are In Sulfur . We hope the information provided has been useful to you. Feel free to contact us if you have any questions or need further assistance. See you next time and don't miss to bookmark.
