How Many Neutrons Does Molybdenum Have
listenit
Apr 02, 2025 · 5 min read
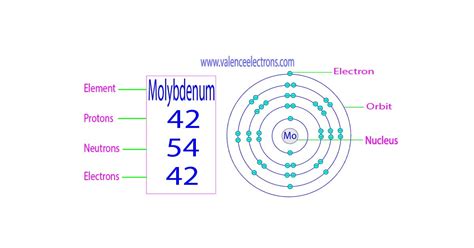
Table of Contents
How Many Neutrons Does Molybdenum Have? A Deep Dive into Isotopes and Nuclear Physics
Molybdenum, a fascinating transition metal with the symbol Mo and atomic number 42, doesn't have a single, definitive answer to the question of "how many neutrons it has." This is because molybdenum exists in nature as a mixture of several isotopes, each possessing a different number of neutrons. Understanding this requires delving into the world of isotopes, nuclear physics, and the implications for various scientific fields.
Understanding Isotopes: The Key to Neutron Count Variation
Before we can answer how many neutrons molybdenum has, we need to grasp the concept of isotopes. Isotopes are atoms of the same element that have the same number of protons (defining the element) but differ in the number of neutrons. This difference in neutron number affects the atom's mass but not its chemical properties. Molybdenum's complexity arises from its numerous stable and radioactive isotopes.
What Defines an Isotope?
The defining characteristics of an isotope are:
- Atomic Number (Z): This represents the number of protons in the nucleus and is unique to each element. For molybdenum, Z = 42.
- Mass Number (A): This is the total number of protons and neutrons in the nucleus (A = Z + N, where N is the number of neutrons). Different isotopes of the same element have different mass numbers.
- Neutron Number (N): This is simply the number of neutrons in the nucleus (N = A - Z).
Molybdenum's Isotopic Abundance: A Mixture of Neutrons
Naturally occurring molybdenum is a mixture of seven stable isotopes, each with a different number of neutrons:
| Isotope | Mass Number (A) | Number of Neutrons (N) | Natural Abundance (%) |
|---|---|---|---|
| ⁹²Mo | 92 | 50 | 14.84 |
| ⁹⁴Mo | 94 | 52 | 9.25 |
| ⁹⁵Mo | 95 | 53 | 15.92 |
| ⁹⁶Mo | 96 | 54 | 16.68 |
| ⁹⁷Mo | 97 | 55 | 9.55 |
| ⁹⁸Mo | 98 | 56 | 24.13 |
| ¹⁰⁰Mo | 100 | 58 | 9.63 |
This table highlights the variability in neutron count. No single number accurately represents the neutron count for molybdenum; it's a weighted average based on the natural abundance of each isotope.
Calculating the Average Neutron Number for Molybdenum
To get a representative average neutron number, we can perform a weighted average calculation using the natural abundance of each isotope:
Average Neutron Number = Σ (Abundance of isotope i * Number of neutrons in isotope i)
Applying this formula to molybdenum's isotopes:
Average Neutron Number ≈ (0.1484 * 50) + (0.0925 * 52) + (0.1592 * 53) + (0.1668 * 54) + (0.0955 * 55) + (0.2413 * 56) + (0.0963 * 58) ≈ 54.17
Therefore, the average number of neutrons in a molybdenum atom is approximately 54.17. It's crucial to remember that this is an average; individual molybdenum atoms will have neutron numbers ranging from 50 to 58.
Radioactive Isotopes of Molybdenum: Expanding the Neutron Spectrum
Beyond the seven stable isotopes, numerous radioactive isotopes of molybdenum have been synthesized. These isotopes have even more diverse neutron numbers, some significantly higher or lower than those found in nature. These radioactive isotopes are valuable tools in various scientific applications, such as:
- Medical Imaging: Certain molybdenum isotopes are used in medical imaging techniques, allowing for diagnosis of various conditions.
- Nuclear Medicine: Some radioactive molybdenum isotopes decay into other useful medical isotopes.
- Industrial Applications: Radioactive molybdenum isotopes are used in various industrial processes such as gauging and tracing.
- Research: They help scientists study nuclear reactions and processes.
These radioactive isotopes often have significantly shorter half-lives, meaning they decay relatively quickly into other elements. This decay process involves the emission of particles, which can alter the number of protons and neutrons in the nucleus, further changing the neutron count.
The Importance of Neutron Number in Molybdenum's Properties
The number of neutrons significantly impacts the properties of molybdenum isotopes. Variations in neutron numbers affect:
- Nuclear Stability: The ratio of protons to neutrons plays a vital role in determining nuclear stability. Isotopes with neutron-to-proton ratios that deviate significantly from the stability zone are often radioactive and undergo decay to achieve stability.
- Mass: The number of neutrons directly contributes to the mass of the atom, which influences its physical properties like density and melting point.
- Nuclear Reactions: The neutron number affects how an isotope interacts in nuclear reactions, such as fission or fusion. This is highly relevant in nuclear power generation and research.
Applications of Molybdenum and its Isotopes
Molybdenum's unique properties, influenced by its varied isotopic composition, make it indispensable in numerous applications:
- Metallurgy: Molybdenum is a crucial alloying element in steel, enhancing its strength and high-temperature properties. This is vital in aerospace, automotive, and other industries demanding high-performance materials.
- Catalysis: Molybdenum compounds act as catalysts in various chemical processes, such as petroleum refining and the production of fertilizers.
- Electronics: Molybdenum is used in electronic components due to its high conductivity and resistance to corrosion.
- Pigments: Molybdenum compounds contribute to vibrant pigments used in paints and plastics.
Conclusion: A Variable Neutron Count Defining a Versatile Element
In conclusion, there's no single answer to how many neutrons molybdenum has. The element exists as a blend of several isotopes, each with a distinct neutron number. The average neutron number is approximately 54.17, but this is only a representative value. The variability in neutron count significantly affects molybdenum's properties and its wide-ranging applications in various fields, from metallurgy and catalysis to nuclear medicine and research. Understanding molybdenum's isotopic composition is crucial for harnessing its unique characteristics and advancing scientific knowledge. Further research into molybdenum isotopes continues to unlock new possibilities and applications. The intricate interplay between protons and neutrons within the molybdenum nucleus continues to intrigue and challenge scientists, highlighting the fascinating world of nuclear physics and its profound impact on our lives.
Latest Posts
Latest Posts
-
What Is 9 Ounces In Cups
Apr 03, 2025
-
Explain Why Chemical Equations Have To Be Balanced
Apr 03, 2025
-
How Do You Write 10 As A Fraction
Apr 03, 2025
-
Why Are Ionic Substances Soluble In Water
Apr 03, 2025
-
How Many Ml In A Dropper
Apr 03, 2025
Related Post
Thank you for visiting our website which covers about How Many Neutrons Does Molybdenum Have . We hope the information provided has been useful to you. Feel free to contact us if you have any questions or need further assistance. See you next time and don't miss to bookmark.
