How Is A Mixture Different From A Compound
listenit
Apr 05, 2025 · 5 min read
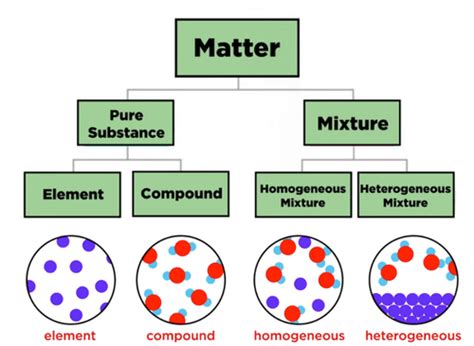
Table of Contents
How is a Mixture Different from a Compound? A Comprehensive Guide
Understanding the fundamental differences between mixtures and compounds is crucial for anyone studying chemistry or related sciences. While both involve combining different substances, the nature of that combination dictates vastly different properties and behaviors. This comprehensive guide will delve into the distinctions between mixtures and compounds, exploring their definitions, characteristics, and providing real-world examples to solidify your understanding. We'll also touch upon the methods used to separate mixtures and the chemical processes involved in the formation and breakdown of compounds.
Defining Mixtures and Compounds: The Core Difference
At its core, the difference between a mixture and a compound lies in the nature of the bonding between the constituent substances.
Mixtures: A mixture is a substance comprising two or more components not chemically bonded. The components retain their individual chemical properties and can be physically separated from one another using various techniques. The composition of a mixture is variable; you can change the ratio of components without altering the fundamental nature of the mixture.
Compounds: A compound, on the other hand, is a substance formed when two or more chemical elements are chemically bonded together. This chemical bonding results in a new substance with entirely new properties, distinct from its constituent elements. The composition of a compound is fixed and defined by its chemical formula. Separating the components of a compound requires chemical reactions, not simple physical processes.
Key Characteristics: A Comparative Analysis
Let's examine the key characteristics that differentiate mixtures from compounds:
| Feature | Mixture | Compound |
|---|---|---|
| Composition | Variable, can be changed | Fixed, defined by chemical formula |
| Bonding | No chemical bonds between components | Chemical bonds between components |
| Properties | Retains properties of its components | Has new properties different from elements |
| Separation | Physically separable | Requires chemical reactions to separate |
| Melting Point | Variable melting point (or range) | Sharp, defined melting point |
| Boiling Point | Variable boiling point (or range) | Sharp, defined boiling point |
Mixture Characteristics Explained:
-
Variable Composition: Think of a bowl of fruit salad. You can add more apples, fewer bananas, or include different fruits entirely, and it's still a fruit salad. The proportions are flexible. This is true for both homogeneous and heterogeneous mixtures.
-
Retention of Properties: In a salt-water mixture, you can still taste the salt and observe the water's properties. The individual components haven't lost their identity.
-
Physical Separation: Simple techniques like filtration, evaporation, distillation, chromatography, and magnetism can separate the components of a mixture.
Compound Characteristics Explained:
-
Fixed Composition: Water (H₂O) always contains two hydrogen atoms for every oxygen atom. Changing this ratio alters the substance's properties entirely; it is no longer water.
-
New Properties: Sodium (Na), a highly reactive metal, and chlorine (Cl), a poisonous gas, combine to form sodium chloride (NaCl), or table salt, a harmless, crystalline compound essential for human life. The properties of the compound are entirely different from its constituent elements.
-
Chemical Separation: Separating the elements of a compound requires chemical processes like electrolysis or chemical decomposition.
Types of Mixtures: Homogeneous vs. Heterogeneous
Mixtures are further classified into two main categories:
Homogeneous Mixtures: These mixtures have a uniform composition throughout. At a macroscopic level, you cannot distinguish the individual components. Examples include saltwater, air (a mixture of gases), and sugar dissolved in water.
Heterogeneous Mixtures: In these mixtures, the composition is not uniform, and you can easily distinguish the individual components. Examples include sand and water, oil and water, and a salad.
Examples of Mixtures and Compounds in Everyday Life
Understanding the difference between mixtures and compounds is easier when you see them in action in everyday life.
Mixtures:
- Air: A homogeneous mixture of gases, primarily nitrogen, oxygen, and argon.
- Seawater: A homogeneous mixture of water and dissolved salts, minerals, and organic matter.
- Milk: A heterogeneous mixture containing water, fat globules, proteins, and carbohydrates.
- Soil: A heterogeneous mixture of mineral particles, organic matter, water, and air.
- Coffee: A heterogeneous mixture (if you have grounds), or homogeneous (if you use instant).
Compounds:
- Water (H₂O): A compound formed from the chemical bonding of hydrogen and oxygen atoms.
- Table Salt (NaCl): A compound formed from the chemical bonding of sodium and chlorine atoms.
- Sugar (C₁₂H₂₂O₁₁): A compound comprised of carbon, hydrogen, and oxygen atoms.
- Carbon Dioxide (CO₂): A compound composed of carbon and oxygen atoms.
- Ammonia (NH₃): A compound containing nitrogen and hydrogen atoms.
Separating Mixtures: A Look at Common Techniques
Several techniques are employed to separate mixtures based on the properties of their components:
-
Filtration: Separates solids from liquids using a filter paper. Think of making coffee – the filter traps the coffee grounds.
-
Evaporation: Separates a dissolved solid from a liquid by evaporating the liquid. This is commonly used to obtain salt from saltwater.
-
Distillation: Separates liquids with different boiling points. This process is used in the production of purified water and alcoholic beverages.
-
Chromatography: Separates components based on their different affinities for a stationary and mobile phase. This is used in various analytical techniques.
-
Magnetic Separation: Separates magnetic materials from non-magnetic materials using a magnet.
Chemical Reactions and Compounds: Formation and Decomposition
Compounds are formed through chemical reactions that involve the rearrangement of atoms and the formation of chemical bonds. These reactions often involve energy changes, such as the release of heat (exothermic) or the absorption of heat (endothermic).
The breakdown of compounds into their constituent elements or simpler compounds also requires chemical reactions. These reactions can be induced by various means such as heating, electrolysis, or reaction with other chemicals.
For example, the electrolysis of water breaks down water (H₂O) into hydrogen (H₂) and oxygen (O₂) gases. This requires an electrical current to provide the necessary energy to break the chemical bonds.
Conclusion: Mastering the Distinction
The difference between mixtures and compounds hinges on the fundamental concept of chemical bonding. Mixtures are physical combinations of substances, while compounds are formed through chemical bonding, resulting in new substances with distinct properties. Understanding this core difference, along with the associated characteristics and separation techniques, is essential for comprehending basic chemistry and its applications in various fields. From everyday substances to complex chemical reactions, the concepts of mixtures and compounds provide a foundation for understanding the world around us. By applying this knowledge, you can better interpret the properties of substances and appreciate the intricacies of chemical processes.
Latest Posts
Latest Posts
-
Hiw Many Ounces In A Pint
Apr 05, 2025
-
How Many Kilograms In 185 Pounds
Apr 05, 2025
-
How Many Right Angles Does Rectangle Have
Apr 05, 2025
-
Write A Chemical Equation For Cellular Respiration
Apr 05, 2025
-
5 Times The Square Root Of 5
Apr 05, 2025
Related Post
Thank you for visiting our website which covers about How Is A Mixture Different From A Compound . We hope the information provided has been useful to you. Feel free to contact us if you have any questions or need further assistance. See you next time and don't miss to bookmark.
