Difference Between Valence Bond Theory And Molecular Orbital Theory
listenit
Apr 05, 2025 · 5 min read
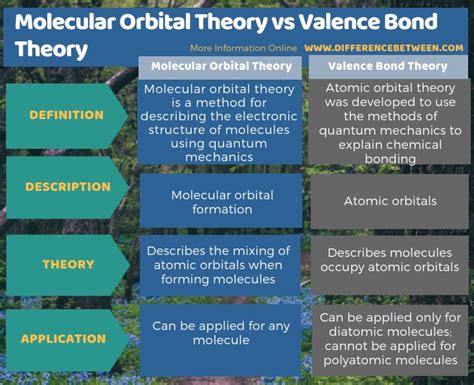
Table of Contents
Delving Deep: Valence Bond Theory vs. Molecular Orbital Theory
Understanding chemical bonding is fundamental to chemistry. Two prominent theories explain how atoms bond to form molecules: Valence Bond Theory (VBT) and Molecular Orbital Theory (MOT). While both aim to describe the same phenomenon, they employ drastically different approaches and offer unique insights into molecular structure and properties. This article will delve into the core differences between VBT and MOT, exploring their strengths, weaknesses, and areas of applicability.
Valence Bond Theory: A Localized Approach
Valence Bond Theory, a cornerstone of introductory chemistry, describes chemical bonds as the overlap of atomic orbitals. It posits that a covalent bond forms when two atoms approach each other, and their valence electrons occupying atomic orbitals interact, creating a region of high electron density between the nuclei. This overlap concentrates electron density, leading to an attractive force that holds the atoms together.
Key Concepts of VBT:
-
Atomic Orbitals: VBT utilizes the atomic orbitals of individual atoms as the foundation for bond formation. These are the familiar s, p, d, and f orbitals described by the quantum mechanical model of the atom.
-
Orbital Overlap: The strength of a bond is directly related to the extent of orbital overlap. Greater overlap results in stronger, shorter bonds. This explains the preference for specific bond geometries – atoms arrange themselves to maximize orbital overlap.
-
Hybridization: To explain the geometry of molecules that contradict simple atomic orbital overlap, VBT introduces the concept of hybridization. Hybridization is the mixing of atomic orbitals to form new hybrid orbitals with different shapes and energies. Common hybrid orbitals include sp, sp², and sp³. This allows us to account for the tetrahedral geometry of methane (CH₄), for example.
-
Resonance: When a single Lewis structure cannot adequately represent the bonding in a molecule, VBT employs the concept of resonance. Resonance structures are different Lewis structures that can be drawn for a molecule, and the true structure is a hybrid of these contributors. This is crucial for understanding molecules like benzene.
-
Limitations of VBT: While VBT offers a simple and intuitive picture of bonding, it has limitations. It struggles to accurately describe molecules with unpaired electrons (radicals) or molecules with delocalized electrons. It also fails to adequately explain the magnetic properties of certain molecules. Furthermore, it doesn't account for the excited states of molecules effectively.
Molecular Orbital Theory: A Delocalized Approach
Molecular Orbital Theory (MOT) takes a more sophisticated approach. It abandons the notion of localized atomic orbitals and instead proposes that when atoms combine, their atomic orbitals combine to form molecular orbitals that encompass the entire molecule. These molecular orbitals can be bonding (lower in energy than the atomic orbitals) or antibonding (higher in energy).
Core Concepts of MOT:
-
Linear Combination of Atomic Orbitals (LCAO): MOT uses the LCAO method to construct molecular orbitals. This involves mathematically combining atomic orbitals of the same symmetry to generate molecular orbitals. The number of molecular orbitals formed equals the number of atomic orbitals combined.
-
Bonding and Antibonding Orbitals: Bonding molecular orbitals concentrate electron density between nuclei, leading to attraction and bond formation. Antibonding orbitals have nodes (regions of zero electron density) between nuclei, resulting in repulsion.
-
Bond Order: The bond order is a measure of the number of bonds between two atoms. It's calculated as (number of electrons in bonding orbitals - number of electrons in antibonding orbitals) / 2. A higher bond order indicates a stronger, shorter bond.
-
Delocalization: MOT excels at describing delocalized electrons, where electrons are spread over multiple atoms. This is particularly relevant in conjugated systems like benzene, where the pi electrons are delocalized across the entire ring.
-
Molecular Orbital Diagrams: Molecular orbital diagrams are visual representations that show the energy levels of molecular orbitals and the occupation of electrons. These diagrams are crucial for predicting molecular properties like bond order, magnetic properties (paramagnetism or diamagnetism), and bond strength.
-
Strengths of MOT: MOT's strength lies in its ability to handle delocalized electrons, predict magnetic properties, and accurately describe excited states of molecules. It provides a more comprehensive picture of bonding, particularly in complex systems.
-
Limitations of MOT: While powerful, MOT can be computationally intensive, especially for large molecules. The mathematical complexity can make it less intuitive than VBT for beginners.
Head-to-Head Comparison: VBT vs. MOT
| Feature | Valence Bond Theory (VBT) | Molecular Orbital Theory (MOT) |
|---|---|---|
| Approach | Localized atomic orbitals | Delocalized molecular orbitals |
| Bond Formation | Overlap of atomic orbitals | Combination of atomic orbitals |
| Bond Description | Localized electron density between atoms | Delocalized electron density across molecule |
| Hybridization | Explains molecular geometry using hybrid orbitals | Not directly used, but implied in orbital mixing |
| Resonance | Uses resonance structures to describe delocalization | Accounts for delocalization naturally |
| Delocalization | Explains delocalization imperfectly | Explains delocalization effectively |
| Magnetic Properties | Less accurate | More accurate |
| Computational Complexity | Relatively simple | Can be computationally intensive |
| Intuitiveness | More intuitive for beginners | Less intuitive for beginners |
| Excited States | Less accurate descriptions | Accurate descriptions |
Which Theory to Use?
The choice between VBT and MOT depends on the specific application and the complexity of the molecule being studied.
-
VBT is suitable for simple molecules where localized bonding is a reasonable approximation. It provides a simple and intuitive understanding of bond formation and molecular geometry, making it ideal for introductory chemistry courses.
-
MOT is necessary for molecules with delocalized electrons, such as conjugated systems and transition metal complexes. Its ability to accurately predict magnetic properties and excited states makes it invaluable in advanced chemical studies.
Beyond the Basics: Advanced Concepts and Applications
Both VBT and MOT have been extended and refined over time to address more complex scenarios. For example:
-
Advanced VBT methods incorporate configuration interaction and other techniques to improve accuracy.
-
Advanced MOT methods include density functional theory (DFT), which is a powerful computational tool used to predict molecular properties.
Both theories continue to be vital tools in modern chemistry, providing complementary perspectives on the intricate world of chemical bonding. Choosing the appropriate theory hinges upon the specific chemical system and the depth of understanding required. While VBT provides a simplified and easily grasped initial comprehension, MOT is crucial for comprehensive analysis and prediction in intricate chemical systems. Their combined application provides a comprehensive view of chemical bonding and molecular structure.
Latest Posts
Latest Posts
-
Solve The Equation H 9 7
Apr 06, 2025
-
An Element Is Determined By The Number Of
Apr 06, 2025
-
What Is 5 Percent Of 50
Apr 06, 2025
-
What Layer Of The Atmosphere Does The Weather Occur
Apr 06, 2025
-
What Is The Difference Between Natural Selection And Artificial Selection
Apr 06, 2025
Related Post
Thank you for visiting our website which covers about Difference Between Valence Bond Theory And Molecular Orbital Theory . We hope the information provided has been useful to you. Feel free to contact us if you have any questions or need further assistance. See you next time and don't miss to bookmark.
