Ca Oh 2 Strong Or Weak
listenit
Apr 01, 2025 · 5 min read
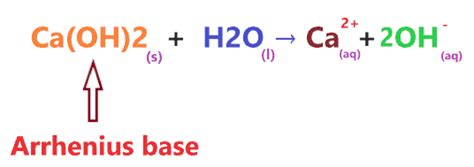
Table of Contents
Ca(OH)₂: Strong or Weak Base? Understanding its Properties and Applications
Calcium hydroxide, Ca(OH)₂, commonly known as slaked lime or hydrated lime, is a widely used chemical compound with significant industrial and environmental applications. A crucial aspect of understanding its behavior and uses lies in classifying it as a strong or weak base. This comprehensive article will delve into the properties of Ca(OH)₂, explore its ionization in water, and ultimately determine its strength as a base. We will also examine its various applications, highlighting how its basicity plays a crucial role in its effectiveness.
Defining Strong and Weak Bases
Before classifying Ca(OH)₂, it's essential to define the terms "strong base" and "weak base." A strong base is a base that completely dissociates (ionizes) into its constituent ions in an aqueous solution. This means that essentially all of the hydroxide ions (OH⁻) are released into the solution. In contrast, a weak base only partially dissociates, meaning a significant portion remains undissociated in solution, resulting in a lower concentration of hydroxide ions. This difference significantly impacts the pH and reactivity of the solutions.
The Dissociation of Calcium Hydroxide
Calcium hydroxide dissolves in water according to the following equilibrium reaction:
Ca(OH)₂(s) ⇌ Ca²⁺(aq) + 2OH⁻(aq)
While this equation suggests a reversible reaction, implying incomplete dissociation, the reality is more nuanced. While not completely dissociated, Ca(OH)₂ exhibits a high degree of dissociation in water. This is due to the relatively high solubility of calcium hydroxide, compared to many other metal hydroxides. The solubility product constant (Ksp) for Ca(OH)₂ is relatively high, indicating a substantial concentration of Ca²⁺ and OH⁻ ions in a saturated solution.
Ksp and the Degree of Dissociation
The solubility product constant, Ksp, is the equilibrium constant for the dissolution of a sparingly soluble ionic compound. For Ca(OH)₂, a higher Ksp value signifies greater solubility and, consequently, a higher concentration of ions in solution. Although Ca(OH)₂ isn't exceptionally soluble, its Ksp is sufficiently high to classify it as a strong base in practical terms. The actual degree of dissociation will depend on factors such as temperature and the presence of other ions in solution. However, the vast majority of Ca(OH)₂ molecules that dissolve will dissociate into ions.
Comparing Ca(OH)₂ to Weak Bases
Let's contrast Ca(OH)₂ with a classic example of a weak base, ammonia (NH₃). Ammonia reacts with water in an equilibrium reaction:
NH₃(aq) + H₂O(l) ⇌ NH₄⁺(aq) + OH⁻(aq)
The equilibrium constant for this reaction, Kb, is much smaller than the Ksp of Ca(OH)₂. This signifies that only a small fraction of ammonia molecules will accept a proton from water and generate hydroxide ions. The resulting hydroxide ion concentration is significantly lower compared to a Ca(OH)₂ solution of comparable concentration. This substantial difference in hydroxide ion concentration directly translates to a difference in pH.
Practical Considerations and Applications
While the theoretical distinction might seem subtle, the practical implications of Ca(OH)₂'s strong basicity are profound. Its high concentration of hydroxide ions makes it an excellent choice for numerous applications:
1. pH Adjustment and Neutralization:
Ca(OH)₂'s strong basicity allows it to effectively neutralize acidic solutions. This is crucial in various industrial processes and wastewater treatment where acidic byproducts need to be neutralized to meet environmental regulations. Its ability to rapidly increase the pH makes it a highly efficient neutralizing agent.
2. Construction and Building Materials:
Lime, in both its quicklime (CaO) and hydrated lime (Ca(OH)₂) forms, is a cornerstone material in the construction industry. It’s used in mortar, plaster, and concrete, where its basicity contributes to setting and hardening processes. The reaction with carbon dioxide in the air further enhances its strength and durability.
3. Water Treatment:
Ca(OH)₂ plays a critical role in water treatment. It's used to adjust the pH of water, to soften hard water by removing magnesium and calcium ions, and as a flocculant to aid in removing impurities from water. Its strong basicity is essential for these processes to be effective.
4. Agriculture and Soil Amendment:
Calcium hydroxide is used in agriculture to adjust soil pH. Acidic soils can hinder plant growth, and the application of Ca(OH)₂ helps raise the pH to an optimal range for better plant health and nutrient uptake. Its strong basicity is vital in neutralizing the acidity.
5. Chemical Industry:
Ca(OH)₂ serves as a reactant or reagent in numerous chemical processes. Its strong basicity makes it useful in various syntheses, including the production of other calcium compounds.
6. Food Industry (indirectly):
While not directly added to food, Ca(OH)₂ plays an indirect role in food processing through its applications in water treatment and sugar refining. The purified water and refined sugar benefit from the use of Ca(OH)₂ in earlier stages.
Safety Precautions
It's crucial to emphasize that while Ca(OH)₂ is a valuable compound, it's also corrosive and can cause skin and eye irritation. Appropriate safety precautions, including the use of protective gear, should always be taken when handling calcium hydroxide.
Conclusion: Ca(OH)₂ - A Strong Base in Practice
While technically not fully dissociating to 100%, calcium hydroxide, Ca(OH)₂, demonstrates a high degree of dissociation in water, leading to a significant concentration of hydroxide ions. Its high Ksp value and its practical applications demonstrate its strong base characteristics. Its ability to effectively neutralize acids, adjust pH, and participate in various chemical and industrial processes all stem from its strong basicity. Therefore, despite the nuances of its equilibrium reaction, Ca(OH)₂ is justifiably classified as a strong base in practical terms, owing to its substantial impact on pH and its wide range of highly effective applications across diverse industries. The high concentration of OH⁻ ions released into the solution is the key determinant in its practical classification as a strong base, despite not achieving 100% dissociation. This characteristic differentiates it significantly from weak bases such as ammonia, which exhibit only partial ionization.
Latest Posts
Latest Posts
-
The Conjugate Base Of Hso4 Is
Apr 03, 2025
-
Examples Of Gay Lussacs Law In Real Life
Apr 03, 2025
-
Law Of Sines And Law Of Cosines Word Problems
Apr 03, 2025
-
18 Is What Percent Of 36
Apr 03, 2025
-
Sin 2x 1 Cos 2x 2
Apr 03, 2025
Related Post
Thank you for visiting our website which covers about Ca Oh 2 Strong Or Weak . We hope the information provided has been useful to you. Feel free to contact us if you have any questions or need further assistance. See you next time and don't miss to bookmark.
