Atoms That Have Gained Or Lost Electrons
listenit
Apr 02, 2025 · 6 min read
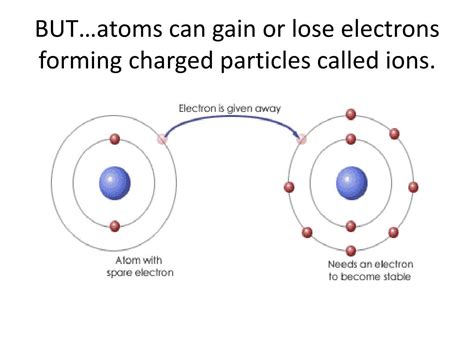
Table of Contents
Atoms That Have Gained or Lost Electrons: Ions and Their Impact
Atoms are the fundamental building blocks of matter, the tiny particles that make up everything around us. But atoms aren't always content to exist in their neutral state. Often, they engage in a crucial process: the transfer or sharing of electrons. When an atom gains or loses electrons, it transforms into an ion, a charged particle with profound implications for chemistry, physics, and even our everyday lives. This article delves deep into the fascinating world of ions, exploring their formation, properties, and significance.
Understanding Atomic Structure: The Foundation of Ion Formation
Before we delve into the intricacies of ion formation, it's crucial to understand the basic structure of an atom. An atom consists of a central nucleus containing positively charged protons and neutral neutrons. Surrounding the nucleus is a cloud of negatively charged electrons that orbit in specific energy levels or shells. The number of protons in an atom's nucleus defines its atomic number, which uniquely identifies the element. In a neutral atom, the number of protons equals the number of electrons, resulting in a net charge of zero.
The Role of Valence Electrons
The electrons in the outermost shell, known as valence electrons, play a pivotal role in chemical bonding and ion formation. These electrons are loosely held and are most likely to be involved in interactions with other atoms. Atoms tend to strive for a stable electron configuration, often achieving this by gaining, losing, or sharing valence electrons to attain a full outermost shell—a state often referred to as achieving a noble gas configuration. This fundamental drive for stability underlies the formation of ions.
Ion Formation: Gaining and Losing Electrons
The process of an atom gaining or losing electrons is called ionization. This process fundamentally alters the atom's charge, transforming it from a neutral entity into an ion.
Cations: Positively Charged Ions
When an atom loses one or more electrons, it becomes positively charged. These positively charged ions are called cations. The loss of electrons occurs because the atom's positive nuclear charge is stronger than the attractive force holding its valence electrons. This often happens with atoms that have relatively few valence electrons, making it energetically favorable to lose them rather than gain more to fill their outer shell.
Example: Sodium (Na) has one valence electron. It readily loses this electron to achieve a stable electron configuration similar to neon (Ne). This results in the formation of a sodium cation, Na⁺.
Anions: Negatively Charged Ions
Conversely, when an atom gains one or more electrons, it becomes negatively charged. These negatively charged ions are called anions. The gain of electrons occurs because the atom's attraction for electrons is stronger than the repulsion from the existing electrons. Atoms with nearly full outer shells are more likely to gain electrons to complete the shell.
Example: Chlorine (Cl) has seven valence electrons. It readily gains one electron to complete its outer shell and achieve a stable configuration similar to argon (Ar). This results in the formation of a chloride anion, Cl⁻.
Factors Influencing Ion Formation
Several factors influence an atom's tendency to gain or lose electrons:
-
Electronegativity: This property describes an atom's ability to attract electrons towards itself in a chemical bond. Atoms with high electronegativity tend to gain electrons, forming anions, while atoms with low electronegativity tend to lose electrons, forming cations.
-
Ionization Energy: This is the energy required to remove an electron from a neutral atom. Atoms with low ionization energies readily lose electrons, forming cations.
-
Electron Affinity: This is the energy change that occurs when an electron is added to a neutral atom. Atoms with high electron affinities readily gain electrons, forming anions.
Properties of Ions: A Marked Difference from Neutral Atoms
Ions exhibit significantly different properties compared to their neutral atom counterparts. These differences stem from the change in the atom's net charge and electron configuration:
-
Charge: The most significant difference is the presence of a net electrical charge. This charge dramatically affects how ions interact with other particles, including other ions, molecules, and electromagnetic fields.
-
Size: Ions often have different sizes compared to their neutral atoms. Cations are typically smaller than their parent atoms because the loss of electrons reduces electron-electron repulsion, allowing the remaining electrons to be pulled closer to the nucleus. Conversely, anions are usually larger than their parent atoms because the addition of electrons increases electron-electron repulsion, causing the electrons to spread out further.
-
Chemical Reactivity: Ions are highly reactive due to their charge. The electrostatic attraction or repulsion between ions plays a major role in driving chemical reactions and the formation of ionic compounds.
-
Physical Properties: The physical properties of ionic compounds, such as melting point, boiling point, and conductivity, are significantly different from those of their constituent elements. Ionic compounds typically have high melting and boiling points due to the strong electrostatic attraction between oppositely charged ions. They also conduct electricity when molten or dissolved in water because the ions become mobile and can carry charge.
The Significance of Ions: Applications Across Diverse Fields
The importance of ions extends far beyond the realm of theoretical chemistry. They play crucial roles in numerous processes and applications:
Biological Systems: Ions and Life
Ions are essential for life. Many biological processes rely heavily on the presence and movement of ions across cell membranes. For instance, nerve impulses are transmitted through the controlled movement of sodium (Na⁺) and potassium (K⁺) ions. Calcium (Ca²⁺) ions are critical for muscle contraction and other cellular processes. The proper balance of various ions is crucial for maintaining homeostasis within living organisms.
Electrochemistry: Harnessing Ion Movement
Electrochemistry utilizes the movement of ions to generate electricity or perform chemical reactions. Batteries, fuel cells, and electrochemical sensors all rely on the flow of ions to function. Electroplating, a process used to coat metal objects with a thin layer of another metal, also involves ion movement.
Industrial Applications: From Metal Extraction to Water Treatment
Ions have various industrial applications. In metallurgy, the extraction of metals from their ores often involves manipulating the ionic forms of the metals. Water treatment processes frequently involve the removal or addition of ions to control water quality. In the semiconductor industry, ions are employed in doping processes to alter the electrical properties of materials.
Medical Applications: Diagnostics and Treatment
Ions play significant roles in medical applications. Electrocardiograms (ECGs) measure electrical activity in the heart, which is driven by the movement of ions. Electrolyte imbalances, involving abnormal concentrations of ions in the body, can lead to various medical conditions. Medical imaging techniques often use ionic contrast agents to improve image visibility.
Conclusion: The Ubiquitous Influence of Ions
Ions, formed through the gain or loss of electrons, are fundamental entities in the world of chemistry and beyond. Their unique properties and reactivity make them indispensable in countless natural and technological processes. From the intricate workings of biological systems to the sophisticated applications in various industries, ions demonstrate their pervasive and profound influence on our world. Understanding the behavior and properties of ions is critical for advancements in diverse fields, including medicine, materials science, and energy technologies. The continued exploration of these charged particles promises exciting discoveries and innovative applications in the years to come.
Latest Posts
Latest Posts
-
500 Ml Of Water Is How Many Ounces
Apr 03, 2025
-
What Is 9 Ounces In Cups
Apr 03, 2025
-
Explain Why Chemical Equations Have To Be Balanced
Apr 03, 2025
-
How Do You Write 10 As A Fraction
Apr 03, 2025
-
Why Are Ionic Substances Soluble In Water
Apr 03, 2025
Related Post
Thank you for visiting our website which covers about Atoms That Have Gained Or Lost Electrons . We hope the information provided has been useful to you. Feel free to contact us if you have any questions or need further assistance. See you next time and don't miss to bookmark.
