All Atoms Of The Same Element Have
listenit
Apr 02, 2025 · 6 min read
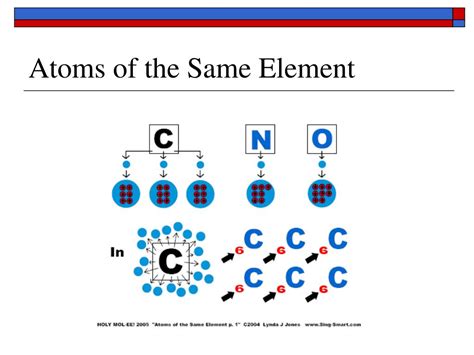
Table of Contents
All Atoms of the Same Element Have: Delving into Atomic Structure and Isotopes
The fundamental building blocks of matter, atoms, are incredibly fascinating entities. A common question that arises when studying chemistry is: "All atoms of the same element have...?" The answer is more nuanced than a simple "the same number of protons." While this is a crucial part of the answer, understanding the full picture requires exploring the intricacies of atomic structure, isotopes, and their implications.
The Defining Characteristic: Atomic Number and Protons
The most fundamental property that defines an element is its atomic number. This number represents the number of protons found in the nucleus of an atom of that element. All atoms of the same element invariably possess the same atomic number, meaning they have the identical number of protons. This is the bedrock of the periodic table's organization, where elements are arranged in order of increasing atomic number. Hydrogen (H), with one proton, occupies the first position, followed by helium (He) with two protons, and so on. This unchanging proton count is what fundamentally differentiates one element from another. For example, all gold atoms have 79 protons; any atom with a different number of protons is simply not gold.
The Significance of Protons in Determining Chemical Properties
The number of protons dictates the element's chemical properties. The protons in the nucleus determine the positive charge of the nucleus, which, in turn, dictates the number of electrons an atom will possess in its neutral state. These electrons reside in energy levels or shells surrounding the nucleus and are responsible for the atom's interaction with other atoms, forming chemical bonds and determining its reactivity. Therefore, since all atoms of the same element have the same number of protons, they inherently exhibit the same fundamental chemical behavior. This consistency is crucial to understanding chemical reactions and predicting the outcomes of chemical processes.
Neutrons: The Variable Component
While protons define the element, the number of neutrons in an atom's nucleus is not as rigidly fixed. Atoms of the same element can have varying numbers of neutrons, giving rise to isotopes.
Isotopes: Variations on a Theme
Isotopes are atoms of the same element that have the same number of protons but different numbers of neutrons. This means they have the same atomic number but different mass numbers. The mass number is the total number of protons and neutrons in the nucleus. For instance, carbon-12 (¹²C) has six protons and six neutrons, while carbon-14 (¹⁴C) has six protons and eight neutrons. Both are isotopes of carbon, exhibiting similar chemical properties due to their identical number of protons, but differing slightly in their physical properties because of the differing neutron count.
The Implications of Varying Neutron Numbers
The differing neutron numbers in isotopes can have significant effects:
- Mass: Isotopes with more neutrons have a greater mass. This mass difference is the basis of techniques like mass spectrometry, which is used to identify and quantify different isotopes within a sample.
- Stability: Some isotopes are stable, meaning their nuclei are unlikely to decay over time. Others are radioactive, meaning their nuclei are unstable and undergo radioactive decay, emitting particles or energy to become more stable. This decay process can have various applications, including medical imaging and carbon dating. For example, ¹⁴C is radioactive and is used in radiocarbon dating to determine the age of organic materials. Its decay rate is consistent, allowing scientists to estimate the time since the organism died.
- Nuclear Reactions: The neutron count can influence the likelihood of an atom undergoing nuclear reactions, such as fission or fusion. Nuclear power plants, for instance, utilize isotopes of uranium, like Uranium-235, which are particularly prone to fission.
Electrons: The Outermost Players
Although the number of protons defines the element, and the number of neutrons can vary, the number of electrons in a neutral atom is directly related to the number of protons. In a neutral atom, the number of electrons is equal to the number of protons, maintaining electrical neutrality. However, atoms can gain or lose electrons, forming ions.
Ions: Charged Atoms
When an atom loses electrons, it becomes a cation, carrying a positive charge. Conversely, when an atom gains electrons, it becomes an anion, carrying a negative charge. The formation of ions dramatically affects the atom's chemical behavior and its interactions with other atoms. Ionic bonding, a fundamental type of chemical bonding, arises from the electrostatic attraction between oppositely charged ions.
The Role of Electrons in Chemical Bonding
The electrons in the outermost energy level (valence electrons) are primarily responsible for the chemical bonding behavior of an atom. Their arrangement determines the atom's reactivity and the types of bonds it can form. The number of valence electrons is directly linked to the atomic number and therefore consistent for all atoms of a given element. This consistency in valence electron arrangement explains the regularity of chemical properties within a group (vertical column) on the periodic table.
Beyond the Basics: Nuclear Spin and Isotopic Abundance
While the number of protons fundamentally defines the element, and the number of neutrons dictates its isotopic variation, there are additional subtle differences at the subatomic level.
Nuclear Spin: A Quantum Mechanical Property
Each proton and neutron possesses a property called nuclear spin, which is a quantum mechanical property analogous to angular momentum. This spin can be thought of as an intrinsic angular momentum. The overall nuclear spin of an atom is the vector sum of the spins of its constituent protons and neutrons. This nuclear spin can affect the atom's interaction with magnetic fields, a principle exploited in techniques like nuclear magnetic resonance (NMR) spectroscopy. While all atoms of the same element have the same number of protons, the nuclear spin can vary depending on the isotope.
Isotopic Abundance: The Natural Mixture
In nature, most elements exist as a mixture of different isotopes. The isotopic abundance refers to the relative proportion of each isotope present in a naturally occurring sample of the element. For instance, carbon exists primarily as ¹²C (approximately 99%) and ¹³C (approximately 1%), with trace amounts of ¹⁴C. This variation in isotopic abundance doesn't change the fundamental chemical properties of the element but does affect the average atomic mass used in stoichiometric calculations. The average atomic mass accounts for the weighted average of the masses of the different isotopes present in a naturally occurring sample.
Conclusion: The Unified View
In summary, the statement "All atoms of the same element have..." is best answered by acknowledging multiple levels of similarity and variation:
- The same number of protons: This is the defining characteristic of an element, dictating its atomic number and fundamental chemical properties.
- The same number of electrons (in the neutral state): This ensures electrical neutrality and directly influences the atom's chemical behavior.
- Potentially different numbers of neutrons: This accounts for the existence of isotopes, leading to variations in mass, stability, and certain physical properties, but not fundamentally altering the chemical behavior.
Understanding the nuances of atomic structure, including protons, neutrons, and electrons, as well as the concept of isotopes and isotopic abundance, is crucial for a comprehensive grasp of chemistry and related fields like physics and materials science. The consistency of proton number within an element forms the basis of the periodic table and our understanding of chemical reactions, while the variations in neutron number account for the fascinating world of isotopes and their diverse applications.
Latest Posts
Latest Posts
-
What Is 5 6 As A Decimal
Apr 03, 2025
-
The Conjugate Base Of Hso4 Is
Apr 03, 2025
-
Examples Of Gay Lussacs Law In Real Life
Apr 03, 2025
-
Law Of Sines And Law Of Cosines Word Problems
Apr 03, 2025
-
18 Is What Percent Of 36
Apr 03, 2025
Related Post
Thank you for visiting our website which covers about All Atoms Of The Same Element Have . We hope the information provided has been useful to you. Feel free to contact us if you have any questions or need further assistance. See you next time and don't miss to bookmark.
