Which Atoms Are Most Likely To Form Covalent Bonds
listenit
Apr 02, 2025 · 5 min read
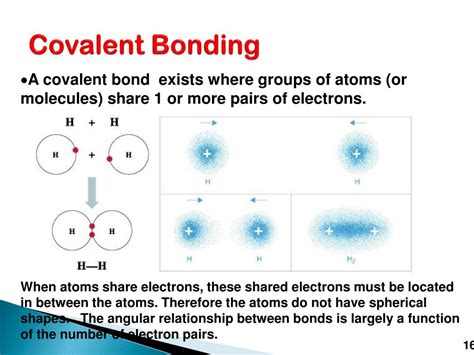
Table of Contents
Which Atoms Are Most Likely to Form Covalent Bonds?
Covalent bonds, the cornerstones of countless molecules in organic and inorganic chemistry, are formed through the sharing of electron pairs between atoms. Understanding which atoms are most likely to form these bonds is crucial for comprehending the structure and properties of matter. This article delves into the factors influencing covalent bond formation, highlighting the atoms and groups of atoms most predisposed to participate in this fundamental chemical interaction.
Electronegativity: The Driving Force Behind Covalent Bonds
The likelihood of an atom forming a covalent bond is intrinsically linked to its electronegativity. Electronegativity is a measure of an atom's ability to attract electrons towards itself in a chemical bond. Atoms with similar electronegativities are more likely to form covalent bonds because neither atom has a strong enough pull to completely steal an electron from the other. Instead, they compromise by sharing electrons.
Understanding the Periodic Trends in Electronegativity
Electronegativity generally increases across a period (from left to right) in the periodic table and decreases down a group (from top to bottom). This trend is directly related to the effective nuclear charge and atomic radius. Elements on the right side of the periodic table, particularly nonmetals, have higher electronegativities than those on the left, the metals.
-
Nonmetals: These elements, located in the upper right-hand corner of the periodic table (excluding the noble gases), readily form covalent bonds due to their high electronegativities. They tend to need only a few more electrons to achieve a stable, full outer electron shell (octet rule). Sharing electrons allows them to achieve this stability more efficiently than transferring electrons.
-
Metals: Metals generally have low electronegativities and tend to lose electrons to form positive ions (cations) rather than sharing them. While some metal-metal bonds can exhibit covalent character, purely covalent bonds between metals are relatively rare.
The Role of the Octet Rule
The octet rule, stating that atoms tend to gain, lose, or share electrons to achieve a full outer shell of eight electrons (like noble gases), is a powerful predictor of covalent bond formation. Atoms strive for this stable electron configuration to minimize their energy. However, it's important to note that the octet rule is a guideline, not an absolute law. There are exceptions, particularly with elements in periods beyond the second row.
Exceptions to the Octet Rule
-
Electron-deficient compounds: Some compounds, like boron trifluoride (BF₃), have fewer than eight electrons around the central atom.
-
Hypervalent compounds: Certain atoms, particularly those in the third period and beyond, can expand their octet and accommodate more than eight electrons in their valence shell. Examples include phosphorus pentachloride (PCl₅) and sulfur hexafluoride (SF₆).
-
Odd-electron compounds: Radicals or free radicals possess an odd number of electrons and thus cannot follow the octet rule.
Specific Atoms and Their Tendency to Form Covalent Bonds
Let's explore specific examples of atoms highly prone to forming covalent bonds:
Group 14 (Carbon Group)
-
Carbon (C): The cornerstone of organic chemistry, carbon's ability to form four covalent bonds is unparalleled. Its tetrahedral structure allows for extensive chain and ring formations, resulting in the vast diversity of organic molecules.
-
Silicon (Si), Germanium (Ge), Tin (Sn), Lead (Pb): While less versatile than carbon, these elements also form covalent bonds, albeit with less stability than carbon-carbon bonds. Organosilicon compounds are an important example of covalent bonding involving silicon.
Group 15 (Nitrogen Group)
-
Nitrogen (N): Nitrogen frequently forms three covalent bonds, as seen in ammonia (NH₃) and countless organic nitrogen compounds. It can also form a triple bond with itself in nitrogen gas (N₂).
-
Phosphorus (P), Arsenic (As), Antimony (Sb), Bismuth (Bi): Similar to nitrogen, these elements readily form covalent bonds, though the stability and properties of their bonds vary.
Group 16 (Oxygen Group)
-
Oxygen (O): Oxygen is highly reactive and commonly forms two covalent bonds, as illustrated in water (H₂O) and countless organic oxygen-containing compounds like alcohols and ethers. It can also form double bonds with itself (O₂) and other elements.
-
Sulfur (S), Selenium (Se), Tellurium (Te), Polonium (Po): These elements share similar bonding tendencies to oxygen, although sulfur can form up to six covalent bonds in certain compounds.
Group 17 (Halogens)
- Fluorine (F), Chlorine (Cl), Bromine (Br), Iodine (I), Astatine (At): Halogens are highly electronegative and typically form one covalent bond to achieve a full octet. Many halogen-containing compounds are known.
Hydrogen (H)
Hydrogen is unique. With only one electron, it can form one covalent bond, often with nonmetals, as seen in water (H₂O), methane (CH₄), and ammonia (NH₃).
Predicting Covalent Bond Formation: A Practical Approach
While electronegativity provides a general guideline, several factors influence the actual formation of covalent bonds:
-
Bond energy: Stronger bonds are more stable and thus more likely to form. Bond energy depends on factors such as the size and electronegativity of the atoms involved.
-
Steric factors: The spatial arrangement of atoms can affect bond formation. Bulky groups may hinder bond formation due to steric hindrance.
-
Resonance: The delocalization of electrons in some molecules can strengthen bonds and enhance stability.
-
Formal charge: A molecule will tend to arrange itself to minimize formal charges on its constituent atoms.
Conclusion: A Diverse World of Covalent Bonds
The formation of covalent bonds is a complex phenomenon influenced by various factors. However, understanding the fundamental concepts of electronegativity, the octet rule, and the periodic trends provides a strong foundation for predicting which atoms are most likely to share electrons and form these essential chemical links. The diversity of covalent compounds, ranging from simple diatomic molecules to complex macromolecules, underscores the crucial role of covalent bonding in shaping the world around us. This understanding is essential in numerous fields, including chemistry, biology, materials science, and more. Further exploration into the nuances of covalent bonding can reveal deeper insights into the fascinating intricacies of the molecular world.
Latest Posts
Latest Posts
-
How To Find Relative Minimum And Maximum
Apr 03, 2025
-
2 3 Or 3 4 Bigger
Apr 03, 2025
-
Friction Is A Non Conservative Force
Apr 03, 2025
-
What Percent Of 27 Is 18
Apr 03, 2025
-
Whats The Square Root Of 500
Apr 03, 2025
Related Post
Thank you for visiting our website which covers about Which Atoms Are Most Likely To Form Covalent Bonds . We hope the information provided has been useful to you. Feel free to contact us if you have any questions or need further assistance. See you next time and don't miss to bookmark.
