Where On The Periodic Table Are The Nonmetals Located
listenit
Apr 05, 2025 · 6 min read
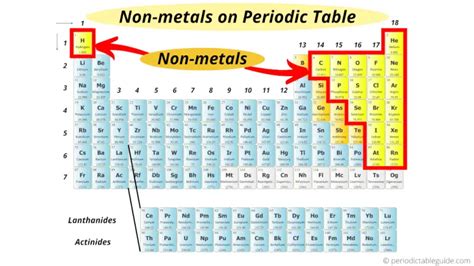
Table of Contents
Where on the Periodic Table are the Nonmetals Located? A Comprehensive Guide
The periodic table, a cornerstone of chemistry, organizes elements based on their atomic structure and properties. Understanding its layout is crucial for grasping the behavior of different elements. This article delves into the location of nonmetals on the periodic table, explaining their characteristics and exploring why they reside where they do. We'll also touch upon the fascinating borderline cases—the metalloids—that blur the lines between metals and nonmetals.
Identifying Nonmetals: A Quick Overview
Before diving into their location, let's establish what defines a nonmetal. Nonmetals are elements that typically lack the characteristic properties of metals. These properties include:
- Poor conductors of heat and electricity: Unlike metals, nonmetals generally resist the flow of heat and electricity.
- Brittle solids: Most nonmetals are brittle, meaning they shatter easily when subjected to stress.
- Low density and melting points: Compared to metals, nonmetals tend to have lower densities and melting points.
- Dull appearance: Nonmetals lack the shiny luster commonly associated with metals.
- Gains electrons easily: In chemical reactions, nonmetals readily gain electrons to achieve a stable electron configuration. This tendency leads to the formation of negative ions (anions).
The Geographic Location of Nonmetals on the Periodic Table
Nonmetals are situated on the right-hand side of the periodic table, with the exception of hydrogen (H), which sits uniquely in the top left corner. This placement isn't arbitrary; it reflects the underlying electron configurations of these elements.
Specifically, the nonmetals occupy:
- Group 17 (Halogens): This group contains highly reactive elements like fluorine (F), chlorine (Cl), bromine (Br), iodine (I), and astatine (At). Their reactivity stems from their tendency to gain a single electron to achieve a stable octet configuration.
- Group 18 (Noble Gases): These elements—helium (He), neon (Ne), argon (Ar), krypton (Kr), xenon (Xe), and radon (Rn)—are exceptionally unreactive due to their full valence electron shells. This makes them chemically inert.
- Upper Right Section: Several other nonmetals reside in the upper right section of the periodic table, including carbon (C), nitrogen (N), oxygen (O), phosphorus (P), sulfur (S), and selenium (Se). These elements exhibit varying degrees of reactivity, depending on their electron configurations and bonding capabilities.
Hydrogen's Unique Position:
Hydrogen's placement is a special case. While it's often placed above Group 1 (alkali metals) due to its single valence electron, it exhibits nonmetallic properties. It's a gas at room temperature, a poor conductor of electricity, and forms covalent bonds, all characteristic of nonmetals. Its unique behavior highlights the complexities of classifying elements neatly.
Why This Location? The Electron Configuration Connection
The positioning of nonmetals isn't coincidental. It's directly linked to the electronic structure of their atoms:
-
Valence Electrons: The number of valence electrons—electrons in the outermost shell—determines an element's chemical behavior. Nonmetals generally have a high number of valence electrons (often 4, 5, 6, or 7). This makes it energetically favorable for them to gain electrons rather than lose them, leading to their characteristic anion formation.
-
Electronegativity: Nonmetals are highly electronegative. Electronegativity measures an atom's ability to attract electrons in a chemical bond. The high electronegativity of nonmetals reflects their strong pull on electrons, further supporting their tendency to gain electrons in reactions.
-
Ionization Energy: Nonmetals have relatively high ionization energies. Ionization energy is the energy needed to remove an electron from an atom. The high ionization energy of nonmetals signifies that it takes significant energy to remove an electron, reinforcing their reluctance to lose electrons.
Metalloids: The In-Between Elements
The boundary between metals and nonmetals isn't sharply defined. A group of elements known as metalloids or semimetals exhibit properties of both metals and nonmetals. These elements are located along a stair-step line that separates metals and nonmetals on the periodic table. Examples of metalloids include boron (B), silicon (Si), germanium (Ge), arsenic (As), antimony (Sb), tellurium (Te), and polonium (Po).
Metalloids possess a blend of characteristics:
- Semiconductivity: Their most notable property is semiconductivity—they conduct electricity better than nonmetals but not as well as metals. This property makes them crucial in the electronics industry.
- Variable Properties: Their properties can vary depending on factors like temperature and pressure.
- Applications: Metalloids find widespread use in semiconductors, alloys, and various other applications.
Exploring Individual Nonmetal Groups in Detail
Let's take a closer look at the major groups of nonmetals:
Halogens (Group 17): Reactive Nonmetals
The halogens are highly reactive because they need only one electron to complete their outermost electron shell. This strong tendency to gain an electron makes them excellent oxidizing agents. Their reactivity decreases as you move down the group (fluorine being the most reactive). Halogens exist as diatomic molecules (e.g., F₂, Cl₂) and form various compounds with other elements.
Noble Gases (Group 18): Inert Elements
The noble gases are unique for their exceptional chemical inertness. Their full valence electron shells make them extremely stable, resulting in minimal reactivity. They are colorless, odorless gases under standard conditions. While traditionally considered inert, some heavier noble gases can form compounds under specific conditions.
Other Nonmetals: A Diverse Group
The remaining nonmetals exhibit a broader range of properties. Oxygen, for example, is crucial for respiration and is a highly reactive element. Nitrogen is a major component of the atmosphere and forms various crucial compounds, including amino acids and proteins. Carbon forms the basis of organic chemistry and exists in diverse forms like diamond and graphite. Sulfur is essential for many biological processes and is used in various industrial applications.
Importance of Nonmetals
Nonmetals play vital roles in our daily lives and the natural world:
-
Biological Importance: Oxygen, nitrogen, carbon, phosphorus, and sulfur are essential for life. They are integral components of biological molecules like DNA, proteins, and carbohydrates.
-
Industrial Applications: Many nonmetals are used extensively in industries. For example, chlorine is used for water purification, nitrogen is used in fertilizers, and carbon is used in materials like plastics and diamonds.
-
Technological Applications: Nonmetals are critical in various technologies. Silicon, for instance, is the foundation of modern electronics.
Conclusion: Mastering the Periodic Table
Understanding the location and characteristics of nonmetals on the periodic table is crucial for grasping the fundamental principles of chemistry. Their placement is directly related to their electron configurations, leading to their distinct properties and behaviors. Knowing their reactivity, electronegativity, and other defining features helps us appreciate their crucial roles in our world, from biological processes to technological advancements. The periodic table serves as a powerful tool, organizing the elements' characteristics and offering a framework for comprehending their chemical interactions. By studying the periodic table diligently, we can unravel the fascinating world of chemistry and its diverse elements.
Latest Posts
Latest Posts
-
Write A Chemical Equation For Cellular Respiration
Apr 05, 2025
-
5 Times The Square Root Of 5
Apr 05, 2025
-
What Are Three Equivalent Fractions For 3 4
Apr 05, 2025
-
How To Find Perpendicular Slope Of A Line
Apr 05, 2025
-
Greatest Common Factor Of 18 And 15
Apr 05, 2025
Related Post
Thank you for visiting our website which covers about Where On The Periodic Table Are The Nonmetals Located . We hope the information provided has been useful to you. Feel free to contact us if you have any questions or need further assistance. See you next time and don't miss to bookmark.
