What Is The Most Reactive Group On The Periodic Table
listenit
Apr 02, 2025 · 5 min read
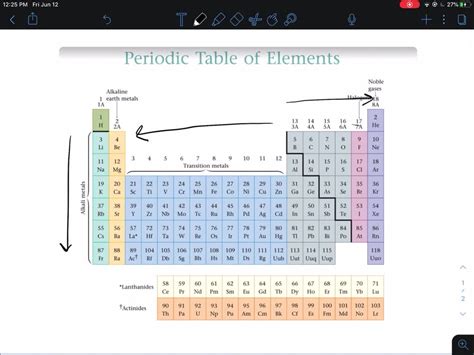
Table of Contents
What is the Most Reactive Group on the Periodic Table?
The periodic table, a cornerstone of chemistry, organizes elements based on their atomic structure and properties. Understanding reactivity is crucial to comprehending chemical behavior, and within the periodic table, certain groups exhibit far more pronounced reactivity than others. While the question of the "most reactive" group requires careful nuance, considering factors like reaction speed and energetic favorability, the alkali metals (Group 1) and the halogens (Group 17) consistently emerge as the strongest contenders. This article delves deep into the reactivity of these groups, exploring the underlying reasons and highlighting specific examples to illustrate their exceptional chemical behavior.
Understanding Reactivity: A Fundamental Concept
Before diving into specific groups, it's important to define reactivity. In chemistry, reactivity refers to the tendency of an element or compound to undergo a chemical reaction, forming new substances. This tendency is governed by several factors including:
- Electron Configuration: Elements strive for stable electron configurations, often achieving this by gaining, losing, or sharing electrons. Those with electron configurations far from stability are generally more reactive.
- Electronegativity: This measures an atom's ability to attract electrons in a chemical bond. Large differences in electronegativity between reacting elements often lead to highly reactive interactions.
- Ionization Energy: The energy required to remove an electron from an atom. Low ionization energies indicate a greater ease of electron loss and hence, higher reactivity.
- Electron Affinity: The energy change when an atom gains an electron. High electron affinities suggest a strong tendency to accept electrons and thus, increased reactivity.
The Alkali Metals (Group 1): Masters of Electron Donation
The alkali metals – lithium (Li), sodium (Na), potassium (K), rubidium (Rb), cesium (Cs), and francium (Fr) – are located in the first column of the periodic table. Their defining characteristic is possessing one valence electron, readily available for donation. This single valence electron is loosely held, resulting in exceptionally low ionization energies. This makes them incredibly reactive, readily participating in reactions that involve electron transfer.
Why are Alkali Metals so Reactive?
- Low Ionization Energy: The single valence electron is easily lost, readily forming a +1 ion. This process is highly energetically favorable, driving the reaction forward.
- Large Atomic Radius: As you move down Group 1, the atomic radius increases. The outermost electron is further from the nucleus and experiences less attraction, making it even easier to remove.
- Electropositive Nature: They readily lose electrons, making them highly electropositive. This contributes to their strong tendency to react with electronegative elements.
Illustrative Reactions of Alkali Metals:
-
Reaction with Water: Alkali metals react violently with water, producing hydrogen gas and a metal hydroxide. The reaction is exothermic (heat-releasing), often resulting in flames and explosions, particularly with the heavier alkali metals like potassium, rubidium, and cesium. For example:
2Na(s) + 2H₂O(l) → 2NaOH(aq) + H₂(g) -
Reaction with Halogens: Alkali metals react vigorously with halogens (Group 17 elements) to form ionic salts. These reactions are highly exothermic and often produce bright flames. For instance:
2Na(s) + Cl₂(g) → 2NaCl(s) -
Reaction with Oxygen: Most alkali metals react readily with oxygen, forming oxides or peroxides. Lithium, for example, forms lithium oxide (Li₂O), while sodium forms sodium peroxide (Na₂O₂).
The Halogens (Group 17): Avid Electron Acceptors
The halogens – fluorine (F), chlorine (Cl), bromine (Br), iodine (I), and astatine (At) – reside in Group 17, possessing seven valence electrons. They exhibit a strong tendency to gain one electron to achieve a stable octet, making them highly reactive non-metals.
Why are Halogens so Reactive?
- High Electron Affinity: Halogens readily accept an electron, forming a -1 ion. This process is energetically favorable, driving their reactions.
- High Electronegativity: Halogens are highly electronegative, strongly attracting electrons in chemical bonds. This enables them to readily pull electrons away from other atoms.
- Small Atomic Radius (for Fluorine and Chlorine): The smaller atomic radii of fluorine and chlorine lead to a stronger attraction for incoming electrons, enhancing their reactivity.
Illustrative Reactions of Halogens:
-
Reaction with Alkali Metals: As mentioned earlier, halogens react vigorously with alkali metals, producing ionic salts (e.g., NaCl, KCl, etc.).
-
Reaction with Hydrogen: Halogens react with hydrogen to form hydrogen halides, which are strong acids in aqueous solutions. For example:
H₂(g) + Cl₂(g) → 2HCl(g) -
Displacement Reactions: A more reactive halogen can displace a less reactive halogen from its salt. For instance, chlorine can displace iodine from potassium iodide:
Cl₂(g) + 2KI(aq) → 2KCl(aq) + I₂(s)
Comparing Reactivity: Alkali Metals vs. Halogens
Determining definitively which group is "most reactive" is challenging. Both alkali metals and halogens display exceptionally high reactivity, but their reactions differ significantly.
- Speed of Reaction: Alkali metals often react explosively fast, especially with water. Halogen reactions are generally fast but less dramatically violent.
- Energy Release: Both groups release considerable energy during reactions, but the specific amounts vary depending on the specific reactants.
- Reaction Types: Alkali metals predominantly undergo oxidation (electron loss), while halogens undergo reduction (electron gain).
Therefore, declaring a single "most reactive" group isn't fully accurate. Both alkali metals and halogens demonstrate exceptionally high reactivity but through different mechanisms. The "most reactive" depends heavily on the specific reaction conditions and the reactants involved.
Other Highly Reactive Groups
While alkali metals and halogens are the most prominent examples, other groups also exhibit significant reactivity:
- Alkaline Earth Metals (Group 2): Possessing two valence electrons, they are less reactive than alkali metals but still readily participate in many reactions.
- Transition Metals: The reactivity of transition metals varies widely depending on the element and its oxidation state. Some, like zinc and iron, are relatively reactive, while others are inert.
- Noble Gases (Group 18): Once considered completely unreactive, recent research has shown that some heavier noble gases can form compounds under specific conditions. Their reactivity remains significantly lower than alkali metals and halogens.
Conclusion: A Spectrum of Reactivity
The periodic table provides a framework for understanding chemical behavior. While the alkali metals and halogens stand out for their exceptional reactivity driven by their electron configurations, the level of reactivity is a spectrum. Many factors, such as ionization energy, electron affinity, electronegativity, and atomic radius, govern how readily elements participate in chemical reactions. The "most reactive" group is therefore context-dependent and not a single, absolute answer. Understanding these underlying principles allows for a deeper appreciation of chemical reactivity and the remarkable diversity of chemical behavior across the periodic table. Further research continues to refine our understanding of these complex interactions, adding to our knowledge of the dynamic world of chemical reactions.
Latest Posts
Latest Posts
-
How Many Cups In Two Gallons
Apr 03, 2025
-
Isotopes Are Atoms Of The Same Element That Have
Apr 03, 2025
-
Is A Dog A Person Place Or Thing
Apr 03, 2025
-
What Is The Fraction Of 78
Apr 03, 2025
-
What Is A Reactant Of Photosynthesis
Apr 03, 2025
Related Post
Thank you for visiting our website which covers about What Is The Most Reactive Group On The Periodic Table . We hope the information provided has been useful to you. Feel free to contact us if you have any questions or need further assistance. See you next time and don't miss to bookmark.
