What Is The Horizontal Row On The Periodic Table
listenit
Apr 04, 2025 · 6 min read
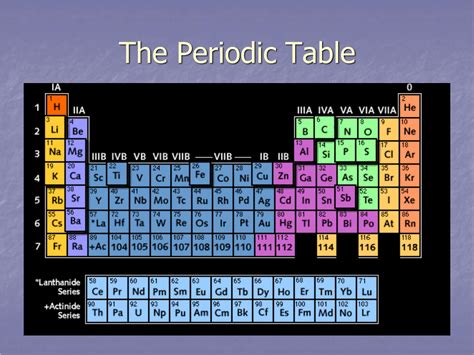
Table of Contents
What is the Horizontal Row on the Periodic Table? Understanding Periods and Their Properties
The periodic table, a cornerstone of chemistry, organizes chemical elements in a structured manner, revealing their properties and relationships. While many focus on the vertical columns (groups), the horizontal rows, known as periods, are equally crucial to understanding the behavior of elements. This comprehensive guide will delve deep into the nature of periods on the periodic table, exploring their significance in determining elemental properties, trends, and their crucial role in predicting chemical reactivity.
Understanding Periods: A Horizontal Journey Through Elements
A period on the periodic table represents a horizontal row. Unlike groups, which share similar chemical properties due to the same number of valence electrons, periods show a trend in properties as you move across them from left to right. This gradual change is due to the sequential increase in the number of protons and electrons within each element's atom. Each period corresponds to the filling of a principal electron shell, also known as an energy level.
The Significance of Electron Shells
The arrangement of electrons within an atom's shells dictates its chemical behavior. Electrons occupy distinct energy levels, and within each level are sub-levels (s, p, d, and f). Periods reflect the filling of these shells. For instance:
- Period 1: Contains only hydrogen (H) and helium (He), filling the 1s subshell.
- Period 2: Elements fill the 2s and 2p subshells.
- Period 3: Elements fill the 3s and 3p subshells.
- Period 4 onwards: The complexity increases, with the inclusion of d and f subshells, leading to the longer periods and the introduction of transition metals and inner transition metals (lanthanides and actinides).
Periodicity of Properties: Trends Across the Rows
The most significant aspect of periods is the periodic trend of properties. As you progress across a period from left to right, several key properties demonstrate a consistent pattern:
1. Atomic Radius: A Shrinking Trend
Atomic radius refers to the size of an atom. Across a period, the atomic radius generally decreases. This is because, while an additional electron shell is added (moving down a group increases atomic radius), within a period, the number of protons in the nucleus increases. The increased positive charge of the nucleus pulls the electrons closer, resulting in a smaller atomic radius.
2. Ionization Energy: An Increasing Climb
Ionization energy is the energy required to remove an electron from a neutral atom. Across a period, ionization energy generally increases. The increased nuclear charge holds the electrons more tightly, making it progressively harder to remove an electron. Exceptions exist due to electron-electron repulsions and the stability of half-filled and filled subshells.
3. Electronegativity: A Pull Towards the Right
Electronegativity measures an atom's ability to attract electrons in a chemical bond. Across a period, electronegativity generally increases. As the nuclear charge increases, the atom's ability to attract electrons from other atoms also increases. Fluorine (F), located in the upper right corner (excluding noble gases), is the most electronegative element.
4. Electron Affinity: The Attraction of an Extra Electron
Electron affinity is the energy change when an atom gains an electron. Across a period, electron affinity generally increases, although the trend is less regular than ionization energy or electronegativity. The added electron is generally attracted more strongly by the increasing nuclear charge. However, electron-electron repulsions can complicate this trend.
5. Metallic Character: A Gradual Transition
Metallic character refers to the properties associated with metals, such as electrical conductivity and malleability. Across a period, metallic character generally decreases. Elements on the left side of the periodic table are typically metals, while those on the right are non-metals. This transition reflects the changes in electron configuration and bonding behavior.
The Special Cases: Noble Gases and Transition Metals
Certain periods exhibit unique characteristics due to specific electron configurations and sublevel filling:
Noble Gases: The Unreactive End
The rightmost column of the periodic table comprises the noble gases (He, Ne, Ar, Kr, Xe, Rn). They are characterized by their full valence electron shells, making them exceptionally stable and unreactive. Their full octet (except for Helium with a duet) explains their inertness, limiting their participation in chemical bonding.
Transition Metals: A Tale of Variable Oxidation States
Periods 4, 5, and 6 feature transition metals, where the d subshells are being filled. These elements exhibit variable oxidation states, meaning they can lose different numbers of electrons to form ions with varying charges. This versatility leads to a wide range of chemical compounds and complex ions.
Inner Transition Metals (Lanthanides and Actinides): Filling the f-Block
The lanthanides and actinides (f-block elements) are placed separately at the bottom of the periodic table. Their unique electronic configurations, involving the filling of the 4f and 5f subshells, lead to distinct chemical properties and similar atomic radii due to the poor shielding effect of the f-electrons.
Period Length and Subshell Filling
The length of each period reflects the number of electrons that can occupy the subshells being filled.
- Periods 1 and 2 (Short Periods): Have only 2 and 8 elements respectively, reflecting the filling of the s and p subshells (2 electrons in the s subshell and 6 electrons in the p subshell).
- Periods 3 and 4 (Long Periods): Have 8 and 18 elements respectively, with the inclusion of 10 d-block elements.
- Periods 5 and 6 (Very Long Periods): Have 18 and 32 elements, respectively, with the inclusion of the 14 f-block elements.
- Period 7 (Incomplete): Remains incomplete, reflecting the ongoing discovery and synthesis of heavier elements, many of which are radioactive and highly unstable.
Predicting Chemical Behavior: The Power of Periods
Understanding the periodic trends within periods is essential for predicting the chemical behavior of elements. For instance, knowing that electronegativity increases across a period allows us to predict the relative bond polarities in compounds formed by elements within that period. Similarly, understanding ionization energy helps in predicting the reactivity of metals and nonmetals.
Conclusion: Periods – Key to Understanding Elemental Properties
The horizontal rows, or periods, on the periodic table are not merely a means of arrangement. They represent a fundamental pattern in the properties of elements. By understanding the trends in atomic radius, ionization energy, electronegativity, electron affinity, and metallic character, we can gain significant insights into the chemical behavior and reactivity of different elements. The periodic table, with its periods and groups, is a powerful tool that unravels the complexities of the chemical world, providing a framework for understanding and predicting the interactions of matter. The sequential filling of electron shells and the resulting trends across periods form the very basis of chemical periodicity. Further research into the intricacies of each period and its elements opens up countless possibilities for exploring new materials and advancing our understanding of the fundamental building blocks of the universe.
Latest Posts
Latest Posts
-
What Is The Energy Transformation That Occurs During Photosynthesis
Apr 04, 2025
-
Number Of Valence Electrons Of Calcium
Apr 04, 2025
-
How Many Neutrons Are In Sulfur
Apr 04, 2025
-
How Many Yards In 180 Inches
Apr 04, 2025
-
Oxidation Number Of Cr In Cr2o72
Apr 04, 2025
Related Post
Thank you for visiting our website which covers about What Is The Horizontal Row On The Periodic Table . We hope the information provided has been useful to you. Feel free to contact us if you have any questions or need further assistance. See you next time and don't miss to bookmark.
