What Is The Compound Name For N2o4
listenit
Apr 02, 2025 · 5 min read
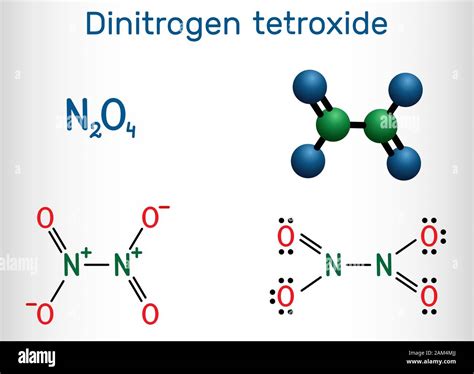
Table of Contents
What is the Compound Name for N₂O₄? A Deep Dive into Dinitrogen Tetroxide
Dinitrogen tetroxide, with the chemical formula N₂O₄, is a fascinating compound with a rich history and diverse applications. This article will explore its properties, synthesis, uses, safety considerations, and environmental impact in detail, providing a comprehensive understanding of this important chemical species.
Understanding the Nomenclature of N₂O₄
Before delving into the specifics, it's crucial to understand why the compound is named dinitrogen tetroxide. Chemical nomenclature follows specific rules to ensure clarity and consistency. In this case:
- Di-: This prefix indicates the presence of two nitrogen atoms.
- Nitrogen: This refers to the element nitrogen (N).
- Tetr-: This prefix indicates the presence of four oxygen atoms.
- Oxide: This suffix signifies that the compound is an oxide, meaning it contains oxygen bonded to another element (nitrogen in this case).
Therefore, dinitrogen tetroxide accurately reflects the chemical composition of N₂O₄, clearly communicating the number of each atom present in the molecule.
Physical and Chemical Properties of Dinitrogen Tetroxide
N₂O₄ exists as a colorless liquid at low temperatures and readily converts to a reddish-brown gas (nitrogen dioxide, NO₂) as temperature increases. This equilibrium between N₂O₄ and NO₂ is a key characteristic, represented by the following reversible reaction:
N₂O₄ ⇌ 2NO₂
The equilibrium shifts towards the formation of NO₂ at higher temperatures and lower pressures. This dynamic equilibrium is crucial in understanding its behavior and applications.
Here's a summary of its key physical and chemical properties:
- Molecular Weight: Approximately 92.01 g/mol
- Melting Point: -11.2 °C (20 °F)
- Boiling Point: 21.2 °C (70 °F)
- Density: 1.45 g/cm³ (liquid at 20 °C)
- Solubility: Soluble in many organic solvents, but reacts with water to form nitric acid and nitrous acid.
This reactivity with water necessitates careful handling and storage procedures. Its strong oxidizing nature makes it a potent reactant in various chemical processes.
Synthesis of Dinitrogen Tetroxide
Several methods can be used to synthesize N₂O₄, primarily focusing on the oxidation of nitrogen oxides or the decomposition of certain nitrogen-containing compounds. While precise laboratory methods are complex, the fundamental principle involves creating conditions favorable for the formation and stabilization of N₂O₄ molecules. One common approach involves the oxidation of nitric oxide (NO) with oxygen (O₂):
2NO + O₂ → 2NO₂
Subsequently, two molecules of NO₂ can dimerize to form N₂O₄ under appropriate conditions (lower temperature and higher pressure). This highlights the importance of controlling temperature and pressure during synthesis to favor the formation of the desired product. Industrial methods often involve more sophisticated processes optimized for large-scale production.
Applications of Dinitrogen Tetroxide
Dinitrogen tetroxide's unique properties lead to a range of applications across various industries, although many involve considerations of its toxicity and reactivity:
1. Rocket Propellant:
One of the most well-known uses of N₂O₄ is as an oxidizer in rocket propellants. When combined with a suitable fuel (such as hydrazine or unsymmetrical dimethylhydrazine), it undergoes a highly exothermic reaction, producing a large amount of thrust. The hypergolic nature of this propellant combination (meaning it ignites spontaneously upon contact) simplifies the engine design and eliminates the need for an ignition system. This characteristic makes it attractive for aerospace applications despite its inherent hazards.
2. Chemical Synthesis:
N₂O₄ serves as a powerful oxidizing agent in various chemical syntheses. Its ability to readily accept electrons makes it a useful reactant for producing nitrated compounds, including explosives. Its reactions often require careful control of reaction conditions to avoid undesirable side products or uncontrolled reactions.
3. Nitration Reactions:
The strong oxidizing nature of N₂O₄ enables its use in nitration reactions. It can introduce nitro groups (-NO₂) into organic molecules, creating compounds with diverse functionalities. This is particularly relevant in the synthesis of certain pharmaceuticals, dyes, and explosives, although its use in these applications requires careful management of safety concerns.
4. Other Applications:
While less common, N₂O₄ finds niche applications in:
- Metal etching: Its oxidizing properties can be utilized for etching and cleaning metal surfaces.
- Polymer chemistry: Some specialized polymer synthesis processes incorporate N₂O₄.
Safety Considerations and Handling of Dinitrogen Tetroxide
N₂O₄ poses significant safety hazards due to its toxicity and reactivity. Appropriate precautions are essential when handling this compound:
- Toxicity: It is highly toxic, both through inhalation and skin contact. Exposure can cause respiratory irritation, pulmonary edema, and even death.
- Reactivity: Its strong oxidizing power makes it highly reactive with many materials, including organic compounds and some metals. This can lead to fires or explosions.
- Corrosivity: It is corrosive to many materials, requiring specialized storage containers and handling equipment.
- Environmental Impact: Release into the environment contributes to air pollution and can have detrimental effects on ecosystems.
Environmental Impact of Dinitrogen Tetroxide
The release of N₂O₄ into the atmosphere contributes to air pollution. Its decomposition products, notably NO₂ and nitric acid, are potent greenhouse gases and air pollutants that contribute to acid rain and smog. Therefore, careful handling and containment are crucial to minimize its environmental impact. Strict regulations and safety protocols govern its industrial use and disposal.
Conclusion: Dinitrogen Tetroxide - A Powerful but Hazardous Compound
Dinitrogen tetroxide (N₂O₄), also known as nitrogen tetroxide, is a fascinating compound with a wide range of applications, primarily driven by its powerful oxidizing properties. Its role in rocket propulsion is particularly significant. However, its inherent toxicity, reactivity, and corrosive nature demand rigorous safety protocols and environmental considerations during its handling, storage, and disposal. The equilibrium between N₂O₄ and NO₂ is a key characteristic that shapes its behavior and applications. Understanding its chemical properties, synthesis methods, uses, and safety concerns is essential for anyone working with or studying this vital yet hazardous chemical. Ongoing research aims to optimize its use while minimizing its potential risks.
Latest Posts
Latest Posts
-
2 3 Or 3 4 Bigger
Apr 03, 2025
-
Friction Is A Non Conservative Force
Apr 03, 2025
-
What Percent Of 27 Is 18
Apr 03, 2025
-
Whats The Square Root Of 500
Apr 03, 2025
-
How Many Feet Is 1 2 Miles
Apr 03, 2025
Related Post
Thank you for visiting our website which covers about What Is The Compound Name For N2o4 . We hope the information provided has been useful to you. Feel free to contact us if you have any questions or need further assistance. See you next time and don't miss to bookmark.
