What Is The Charge On A Potassium Ion
listenit
Apr 03, 2025 · 6 min read
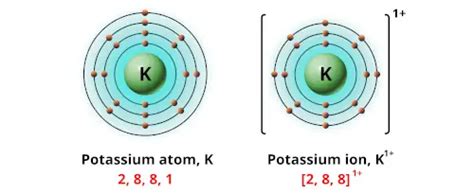
Table of Contents
What is the Charge on a Potassium Ion? A Deep Dive into Ionic Charges and Potassium's Role
Potassium, a crucial element for life, exists in nature not as a lone atom but as an ion – a charged particle. Understanding the charge on a potassium ion is fundamental to comprehending its behavior in biological systems, its chemical reactivity, and its applications in various fields. This article delves into the intricacies of ionic charges, focusing specifically on the potassium ion (K⁺), exploring its formation, properties, and significance.
Understanding Ionic Charges: A Basic Primer
Before diving into the specifics of potassium, let's establish a foundation in ionic charges. Atoms are inherently electrically neutral, possessing an equal number of positively charged protons in their nucleus and negatively charged electrons orbiting it. However, atoms can gain or lose electrons to achieve a more stable electron configuration, a state of lower energy often referred to as a full outer electron shell (octet rule). This process results in the formation of ions.
-
Cations: When an atom loses one or more electrons, it becomes positively charged because it now has more protons than electrons. These positively charged ions are called cations. The magnitude of the positive charge depends on the number of electrons lost. For example, losing one electron results in a +1 charge, losing two electrons results in a +2 charge, and so on.
-
Anions: Conversely, when an atom gains one or more electrons, it becomes negatively charged due to an excess of electrons. These negatively charged ions are called anions. Similar to cations, the magnitude of the negative charge is determined by the number of electrons gained.
The Charge on a Potassium Ion: +1
Potassium (K), an alkali metal located in Group 1 of the periodic table, readily loses one electron to achieve a stable electron configuration resembling that of the noble gas Argon. This electron loss transforms the neutral potassium atom into a potassium ion, denoted as K⁺. The +1 charge signifies the loss of a single negatively charged electron, leaving a net positive charge on the ion.
This tendency to lose an electron and form a +1 ion is a defining characteristic of alkali metals. Their outer electron shell contains only one electron, which is relatively loosely held. This makes it energetically favorable for them to lose this single electron and achieve a stable, filled electron shell.
Why the +1 Charge is Crucial
The +1 charge on the potassium ion is not merely a matter of textbook chemistry; it has profound implications for its behavior and function:
-
Electrochemical Gradients: The positive charge of K⁺ is essential for establishing electrochemical gradients across cell membranes. These gradients are fundamental to nerve impulse transmission, muscle contraction, and many other vital biological processes. The movement of potassium ions across membranes is tightly regulated by specialized ion channels and pumps.
-
Enzyme Activity: Many enzymes require specific ions to function optimally. Potassium ions often play a crucial catalytic role, interacting with enzyme active sites and facilitating biochemical reactions. The +1 charge contributes to these interactions.
-
Osmotic Balance: Potassium ions are involved in maintaining osmotic balance within cells and tissues. This means regulating the distribution of water and maintaining appropriate fluid pressure. The ionic charge influences these interactions.
-
Plant Physiology: Potassium is a vital nutrient for plants. Its +1 charge allows it to interact with negatively charged soil particles and be absorbed by plant roots. It plays a crucial role in stomatal regulation, photosynthesis, and overall plant growth.
Formation of the Potassium Ion: A Detailed Look
The formation of K⁺ involves the ionization energy of potassium. Ionization energy is the energy required to remove an electron from a neutral atom. For potassium, the first ionization energy is relatively low compared to other elements, reflecting the ease with which it loses its outermost electron.
This low ionization energy is explained by the electronic configuration of potassium. Its electron configuration is [Ar] 4s¹. The 4s electron is relatively far from the nucleus and experiences weak attraction, making it easier to remove. Removing this single electron leaves a complete and stable electron shell resembling that of Argon ([Ar]), a noble gas. This stable configuration is the driving force behind potassium's tendency to form a +1 ion.
Potassium Ion's Role in Biology: A Closer Examination
Potassium's +1 charge is inextricably linked to its numerous vital roles in biological systems. Let's delve into some key examples:
1. Nerve Impulse Transmission
The transmission of nerve impulses relies heavily on the movement of potassium ions across neuronal membranes. The resting membrane potential, a crucial factor in nerve excitability, is partly maintained by the higher concentration of potassium ions inside the neuron compared to the extracellular fluid. When a neuron is stimulated, potassium channels open, allowing potassium ions to flow out of the neuron, contributing to the repolarization phase of the action potential. This carefully controlled movement of charged particles underlies the rapid signaling within the nervous system.
2. Muscle Contraction
Similar to nerve impulse transmission, muscle contraction is also dependent on the precise regulation of potassium ion movement. The interplay between potassium and other ions like sodium and calcium is crucial for the excitation-contraction coupling process. Changes in potassium ion concentration affect membrane potential, influencing the contractile properties of muscle fibers.
3. Regulation of Heartbeat
Potassium ions play a critical role in maintaining a regular and efficient heartbeat. Imbalances in potassium levels can lead to cardiac arrhythmias. The heart's electrical activity relies on the precise movement of potassium ions across cardiac muscle cell membranes. Appropriate potassium levels are essential for the proper function of the sinoatrial (SA) node, which sets the pace of the heartbeat.
4. Maintaining Fluid Balance
Potassium contributes to the regulation of fluid balance throughout the body. Its distribution across different compartments, such as intracellular and extracellular fluids, is influenced by its +1 charge and its interactions with other ions and water molecules. Maintaining proper potassium levels is essential for maintaining osmotic pressure and preventing fluid imbalances.
Potassium Ion in Other Applications
Beyond its biological importance, the potassium ion has several applications in other areas:
-
Fertilizers: Potassium is a crucial component of many fertilizers because of its role in plant growth. Its +1 charge allows for its absorption by plant roots.
-
Electrolyte solutions: Potassium salts are used in electrolyte solutions for various industrial and medical applications.
-
Chemical reactions: Potassium ions can participate in various chemical reactions as a counterion or a reactant.
Conclusion: The Importance of Understanding Ionic Charge
The +1 charge on the potassium ion is not just a simple fact; it is a fundamental property that dictates its behavior, reactivity, and essential roles in biological systems. This charge is responsible for its ability to establish electrochemical gradients, participate in enzyme catalysis, regulate osmotic balance, and contribute to countless other critical processes. Understanding the charge on ions like potassium is crucial for comprehending their behavior in various settings, from the intricate workings of a living cell to industrial applications. The simplicity of a single positive charge belies the complexity and significance of this essential element's contribution to life and technology. Further research continues to reveal the intricacies of potassium's function, emphasizing the importance of understanding fundamental chemical principles like ionic charge in unlocking deeper insights into natural phenomena and technological advancements.
Latest Posts
Latest Posts
-
What Are The Two Strands Of Dna Held Together By
Apr 04, 2025
-
How Many Atoms Of Oxygen Are In H2o
Apr 04, 2025
-
Do Homologous Chromosomes Have The Same Alleles
Apr 04, 2025
-
What Are Coefficients In A Chemical Equation
Apr 04, 2025
-
What Is Ph Of Normal Rain
Apr 04, 2025
Related Post
Thank you for visiting our website which covers about What Is The Charge On A Potassium Ion . We hope the information provided has been useful to you. Feel free to contact us if you have any questions or need further assistance. See you next time and don't miss to bookmark.
