What Color Light Has The Highest Energy
listenit
Apr 03, 2025 · 5 min read
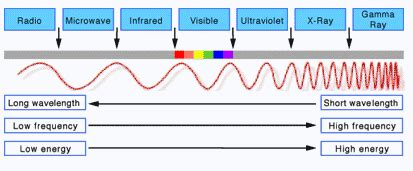
Table of Contents
What Color Light Has the Highest Energy? Unveiling the Secrets of the Electromagnetic Spectrum
The world around us is awash in light, a seemingly simple phenomenon that underpins life itself. But beneath the surface of everyday perception lies a complex tapestry of energy and wavelengths, each with its own unique properties. One of the most fundamental questions regarding light is: what color light has the highest energy? This article delves into the fascinating world of the electromagnetic spectrum, exploring the relationship between light's color, wavelength, frequency, and energy, ultimately answering this crucial question and illuminating the underlying physics.
Understanding the Electromagnetic Spectrum
Before we can pinpoint the highest-energy color of light, it's crucial to grasp the concept of the electromagnetic spectrum. This spectrum encompasses all forms of electromagnetic radiation, ranging from extremely low-energy radio waves to incredibly high-energy gamma rays. Visible light, the portion we can see with our eyes, occupies a tiny sliver within this vast spectrum.
The Components of the Electromagnetic Spectrum:
The electromagnetic spectrum is characterized by its wavelength and frequency, which are inversely proportional.
-
Wavelength: This refers to the distance between two successive peaks (or troughs) of a wave. It's typically measured in nanometers (nm) or angstroms (Å).
-
Frequency: This represents the number of wave cycles that pass a given point per second. It's measured in Hertz (Hz).
The energy of electromagnetic radiation is directly proportional to its frequency and inversely proportional to its wavelength. This crucial relationship is described by the equation: E = hf, where E is energy, h is Planck's constant, and f is frequency.
Visible Light and its Colors:
Visible light, the portion of the spectrum our eyes can detect, comprises a range of wavelengths and frequencies, each corresponding to a specific color. The colors we perceive, arranged from longest wavelength to shortest, are red, orange, yellow, green, blue, indigo, and violet (often remembered by the acronym ROY G. BIV).
The Relationship Between Color, Wavelength, and Energy
Now, let's address the core question: which color of light possesses the highest energy? Since energy is directly proportional to frequency and inversely proportional to wavelength, the color with the shortest wavelength and highest frequency will have the highest energy.
Within the visible light spectrum, violet light has the shortest wavelength and the highest frequency. Consequently, violet light possesses the highest energy among the visible colors.
A Deeper Dive into the Physics:
The energy of a photon (a particle of light) is quantized, meaning it exists in discrete packets. The energy of a single photon is directly proportional to its frequency, as stated in Planck's equation (E = hf). Since violet light has the highest frequency within the visible spectrum, each violet photon carries the most energy.
Beyond Visible Light: Higher Energy Radiation
While violet light holds the title for highest energy within the visible spectrum, it's important to note that significantly higher energy radiation exists beyond the visible range. Ultraviolet (UV) light, X-rays, and gamma rays all possess shorter wavelengths and higher frequencies than violet light, making them far more energetic.
Ultraviolet (UV) Radiation:
UV light lies just beyond violet light in the electromagnetic spectrum. Its higher frequency and shorter wavelength lead to higher energy photons, capable of causing sunburn and damaging DNA. The three types of UV radiation—UVA, UVB, and UVC—differ in their wavelengths and energy levels, with UVC being the most energetic.
X-rays:
X-rays have much shorter wavelengths and even higher frequencies than UV light, resulting in significantly higher energy photons. Their high energy allows them to penetrate soft tissues, making them invaluable in medical imaging.
Gamma Rays:
Gamma rays represent the highest-energy form of electromagnetic radiation. They have extremely short wavelengths and incredibly high frequencies, possessing the highest energy photons in the entire electromagnetic spectrum. Gamma rays are extremely dangerous and can cause severe damage to living tissue.
Practical Applications of High-Energy Light
Understanding the energy levels of different types of light has profound implications across numerous scientific and technological fields.
Medical Applications:
- X-ray imaging: Utilizing the penetrating power of X-rays to visualize internal structures of the body.
- Radiation therapy: Employing high-energy gamma rays to target and destroy cancerous cells.
- Photodynamic therapy: Utilizing specific wavelengths of light to activate photosensitive drugs, targeting and destroying cancerous cells or pathogens.
Industrial Applications:
- Sterilization: Employing UV light to sterilize surfaces and equipment by damaging the DNA of microorganisms.
- Material processing: Utilizing lasers emitting specific wavelengths of light for precise cutting, welding, and other material processing techniques.
- Spectroscopy: Analyzing the absorption and emission spectra of materials to determine their composition and properties.
Scientific Research:
- Astronomy: Studying the electromagnetic radiation emitted by celestial objects to understand their properties and composition.
- Physics: Investigating the fundamental properties of light and its interaction with matter.
- Chemistry: Utilizing spectroscopy and other light-based techniques to analyze chemical reactions and structures.
Conclusion: The Significance of Understanding Light Energy
Understanding the relationship between the color of light, its wavelength, frequency, and energy is fundamental to numerous fields. While violet light holds the highest energy within the visible spectrum, it's crucial to recognize that significantly more energetic forms of electromagnetic radiation exist beyond the visible range. This knowledge underpins countless applications in medicine, industry, and scientific research, shaping our understanding of the universe and our place within it. From medical imaging to astronomical observations, the power of light, and specifically, its energy, continues to revolutionize our world. The seemingly simple question of "what color light has the highest energy?" opens up a vast and fascinating exploration of the electromagnetic spectrum and its profound impact on our lives. Further research into the intricacies of light and its interactions continues to unveil new possibilities and applications, promising a future brimming with innovation driven by the power of light.
Latest Posts
Latest Posts
-
Where Is Most Freshwater On Earth Found
Apr 04, 2025
-
Mars Distance From Earth In Light Years
Apr 04, 2025
-
What Energy Transformation Takes Place During Photosynthesis
Apr 04, 2025
-
What Does A Higher Specific Heat Mean
Apr 04, 2025
-
Question Plane Draw The Skeletal Structures
Apr 04, 2025
Related Post
Thank you for visiting our website which covers about What Color Light Has The Highest Energy . We hope the information provided has been useful to you. Feel free to contact us if you have any questions or need further assistance. See you next time and don't miss to bookmark.
