The Substance That Is Dissolved In A Solution
listenit
Apr 06, 2025 · 6 min read
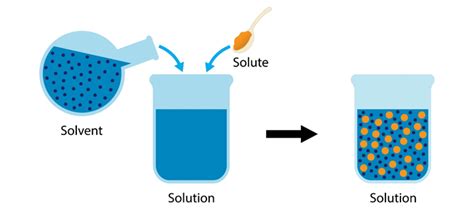
Table of Contents
The Substance That is Dissolved in a Solution: A Deep Dive into Solutes
The world around us is a tapestry woven with solutions. From the salty ocean to the fluids within our bodies, solutions are ubiquitous. Understanding solutions requires grasping the fundamental concept of the solute, the substance being dissolved. This comprehensive article will explore solutes in detail, examining their properties, classifications, and roles in various chemical and biological processes. We'll delve into the fascinating interactions between solutes and solvents, examining the factors that influence solubility and the consequences of these interactions.
What is a Solute?
A solute is a substance that dissolves in a solvent to form a solution. It's the component of a solution that is present in a smaller amount than the solvent. Think of making sweet tea: the sugar (solute) dissolves in the water (solvent) to create a sweet tea solution. The solute is uniformly distributed throughout the solution at the molecular or ionic level, meaning it's not visibly separated from the solvent.
Key Characteristics of Solutes:
- Solubility: This is the crucial property of a solute. It refers to the maximum amount of solute that can dissolve in a given amount of solvent at a specific temperature and pressure. High solubility means the solute readily dissolves, while low solubility means it only dissolves to a limited extent.
- Polarity: The polarity of a solute significantly impacts its solubility. Polar solutes, like sugar and salt, dissolve well in polar solvents like water. This is because polar molecules have regions of positive and negative charge, allowing them to interact with the polar water molecules through dipole-dipole interactions and hydrogen bonding. Nonpolar solutes, like oil and fats, dissolve well in nonpolar solvents like benzene. These interactions are based on weaker London dispersion forces. This principle is often summarized as "like dissolves like."
- Particle Size: The size of solute particles influences the rate of dissolution. Smaller particles dissolve faster than larger ones because they have a greater surface area exposed to the solvent. This is why powdered sugar dissolves faster than a sugar cube.
- Temperature: Temperature affects solubility. For most solid solutes, solubility increases with increasing temperature. However, the solubility of gases in liquids typically decreases with increasing temperature.
- Pressure: Pressure significantly affects the solubility of gases in liquids. Henry's Law states that the solubility of a gas is directly proportional to the partial pressure of the gas above the liquid.
Types of Solutes:
Solutes can be broadly classified based on their chemical nature and behavior in solution:
1. Ionic Solutes:
These solutes are composed of ions, which are charged atoms or molecules. When ionic compounds dissolve in a polar solvent like water, they dissociate into their constituent ions. For example, table salt (NaCl) dissociates into Na⁺ and Cl⁻ ions in water. These ions are then surrounded by water molecules, a process called hydration, which stabilizes them in the solution. Ionic solutes are typically strong electrolytes, meaning they readily conduct electricity in solution. Examples include:
- Sodium chloride (NaCl): Common table salt.
- Potassium hydroxide (KOH): A strong base.
- Calcium chloride (CaCl₂): Used for de-icing roads.
2. Molecular Solutes:
These solutes are composed of molecules, which are electrically neutral groups of atoms. Depending on their polarity, molecular solutes can behave differently in solution. Polar molecular solutes dissolve well in polar solvents through dipole-dipole interactions and hydrogen bonding, while nonpolar molecular solutes dissolve well in nonpolar solvents through London dispersion forces. Examples include:
- Sugar (Sucrose): A polar molecular solute.
- Ethanol (C₂H₅OH): A polar molecular solute, miscible with water.
- Benzene (C₆H₆): A nonpolar molecular solute.
3. Metallic Solutes:
Some metals can dissolve in certain solvents, especially liquid metals. This type of solution is less common than ionic or molecular solutions but is important in certain metallurgical processes. The solubility of metals depends on factors like temperature and the nature of the solvent metal.
4. Gaseous Solutes:
Gases can dissolve in liquids, although their solubility is typically lower than that of solids or liquids. The solubility of a gas in a liquid is affected by temperature and pressure. Examples include:
- Carbon dioxide (CO₂): Dissolved in carbonated beverages.
- Oxygen (O₂): Dissolved in water, crucial for aquatic life.
- Nitrogen (N₂): Dissolved in blood.
The Role of Solutes in Various Processes:
Solutes play crucial roles in numerous natural and industrial processes:
1. Biological Systems:
In biological systems, solutes are essential for various functions. Ions like Na⁺, K⁺, Ca²⁺, and Cl⁻ are crucial for nerve impulse transmission, muscle contraction, and maintaining osmotic balance. Glucose and other sugars serve as energy sources, while amino acids are the building blocks of proteins. Many vital biochemical reactions occur in aqueous solutions, where solutes are dissolved in water.
2. Industrial Processes:
Solutes are central to many industrial processes. In the chemical industry, solutions are used for various reactions, separations, and purifications. Electroplating involves dissolving a metal into a solution and then depositing it onto another surface. In the food industry, solutes are used to add flavor, color, and preservatives.
3. Environmental Science:
The solubility of solutes influences water quality and environmental pollution. The presence of certain solutes can make water unsafe for drinking or harm aquatic life. Understanding solute behavior is crucial for managing pollution and protecting water resources.
Factors Affecting Solubility:
Several factors influence the solubility of a solute in a solvent:
- Temperature: As mentioned earlier, temperature affects solubility differently for solids and gases. Generally, increasing temperature increases the solubility of solids but decreases the solubility of gases.
- Pressure: Pressure primarily affects the solubility of gases. Increased pressure increases the solubility of gases in liquids, as described by Henry's Law.
- Nature of Solute and Solvent: The "like dissolves like" rule is paramount. Polar solutes dissolve well in polar solvents, and nonpolar solutes dissolve well in nonpolar solvents. The strength of intermolecular forces between solute and solvent molecules dictates the solubility.
- Presence of Other Solutes: The presence of other solutes in a solution can affect the solubility of a given solute. This can be due to common ion effects or complexation reactions.
Supersaturated Solutions and Solubility Limits:
A saturated solution contains the maximum amount of solute that can dissolve in a given amount of solvent at a specific temperature and pressure. A supersaturated solution contains more solute than it can theoretically hold at that temperature and pressure. These are often metastable states, meaning they are unstable and can easily revert to a saturated solution by precipitating out the excess solute. Understanding solubility limits is essential in various applications, from crystallization processes to designing pharmaceutical formulations.
Conclusion:
The solute, the substance that dissolves in a solution, is a fundamental concept in chemistry and numerous other scientific disciplines. Its properties, interactions with solvents, and roles in various processes are diverse and multifaceted. Understanding the factors that influence solubility and the behavior of solutes is crucial in various fields, ranging from medicine and environmental science to materials science and industrial chemistry. The exploration of solutes continues to unveil new insights and applications, highlighting their importance in our world. Further research into the intricacies of solute-solvent interactions promises to unlock even more potential in the future. From understanding the delicate balance of ions in our bodies to developing innovative materials and technologies, the study of solutes remains a vital area of scientific inquiry.
Latest Posts
Latest Posts
-
Is Sodium Solid Liquid Or Gas
Apr 06, 2025
-
What Units Are Used To Measure Power
Apr 06, 2025
-
Empirical Formula Of Copper Ii Oxide
Apr 06, 2025
-
Perform The Operation And Write The Result In Standard Form
Apr 06, 2025
-
What Is The Concentration Of Water
Apr 06, 2025
Related Post
Thank you for visiting our website which covers about The Substance That Is Dissolved In A Solution . We hope the information provided has been useful to you. Feel free to contact us if you have any questions or need further assistance. See you next time and don't miss to bookmark.
