Number Of Valence Electrons For Helium
listenit
Apr 03, 2025 · 5 min read
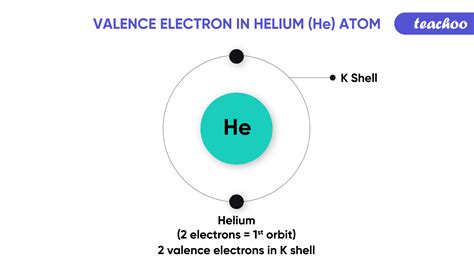
Table of Contents
The Unique Valence Electron Configuration of Helium: A Deep Dive
Helium, the second element on the periodic table, holds a special place in chemistry due to its unique electronic structure. Unlike most other elements, understanding its valence electrons requires a nuanced approach. This article delves deep into the intricacies of helium's valence electron configuration, exploring its implications for chemical bonding and its overall behavior. We'll examine the underlying principles of electron configuration, explore why helium is exceptionally stable, and discuss its role in various scientific applications.
Understanding Valence Electrons
Before focusing specifically on helium, let's establish a foundational understanding of valence electrons. Valence electrons are the electrons located in the outermost shell of an atom. These electrons are the primary players in chemical bonding, determining an element's reactivity and the types of bonds it can form. The number of valence electrons dictates an element's position within the periodic table and its group properties. For example, elements in Group 1 (alkali metals) have one valence electron, while those in Group 18 (noble gases) typically have a full outer shell of valence electrons.
The arrangement of electrons within an atom is governed by quantum mechanics. Electrons occupy specific energy levels or shells, each capable of holding a limited number of electrons. The first shell can hold a maximum of two electrons, the second shell eight, and so on. This arrangement dictates an atom's chemical behavior.
Helium's Electronic Configuration: A Stable Duet
Helium's atomic number is 2, meaning it possesses two protons and, in its neutral state, two electrons. Crucially, these two electrons completely fill its first and only electron shell. This full shell is the key to understanding helium's unique properties. Unlike most elements which aim for an octet (eight valence electrons) for stability, helium achieves stability with a duet—a full first shell containing two electrons.
This stable duet configuration is the reason helium is an inert gas, meaning it is exceptionally unreactive. It has no tendency to gain, lose, or share electrons to achieve a more stable configuration because it already possesses the most stable configuration possible for its electron shell. This lack of reactivity is a defining characteristic of the noble gases, a group that also includes neon, argon, krypton, xenon, and radon.
The Significance of the First Shell
The first electron shell's capacity for only two electrons is a consequence of the quantum mechanical principles governing electron behavior. The first shell consists solely of an s orbital, which can accommodate a maximum of two electrons, each with opposite spin (Pauli Exclusion Principle). This contrasts with subsequent shells, which have both s and p orbitals, allowing for a higher electron capacity.
This fundamental difference between the first shell and others is what makes helium's duet configuration so significant. The complete filling of this innermost shell results in exceptional stability, making helium exceptionally resistant to chemical reactions.
Why Helium Doesn't Form Bonds
The extreme stability of helium's electron configuration explains why it rarely, if ever, participates in chemical bonding. Chemical bonding arises from the tendency of atoms to achieve a more stable electron configuration, typically by filling their outermost electron shell. Since helium already possesses a stable, full shell, it has no energetic incentive to interact with other atoms through electron sharing, donating, or receiving.
This lack of reactivity has profound implications for helium's behavior and applications. It explains why helium is a monatomic gas (existing as individual atoms, not molecules) and its low boiling point, reflecting the weak interatomic forces between helium atoms.
Helium's Role in Science and Technology
Despite its unreactivity, helium's unique properties make it invaluable in numerous scientific and technological applications. Its low density, low boiling point, and inertness are all key attributes exploited in various fields.
1. Cryogenics:
Helium's extremely low boiling point (-268.93°C) allows it to be used as a coolant in cryogenic applications, maintaining extremely low temperatures necessary for superconducting magnets in MRI machines and research involving superconductivity.
2. Leak Detection:
Helium's small atomic size and inert nature enable its use as a leak detection agent. Its ability to penetrate even the tiniest leaks makes it crucial in applications such as detecting leaks in high-vacuum systems.
3. Welding and Arc-Welding:
Helium's inertness protects the weld from oxidation during welding processes, ensuring a high-quality weld. Its thermal conductivity allows it to assist in dissipating heat, improving welding efficiency.
4. Scientific Instrumentation:
Helium's inertness and low reactivity are crucial in various scientific instruments, such as gas chromatography and mass spectrometry. It acts as a carrier gas without interfering with the analysis.
5. Balloons and Airships:
Helium's low density makes it a safer alternative to hydrogen for inflating balloons and airships. Its inertness prevents the risk of combustion.
Comparing Helium to Other Noble Gases
While all noble gases are relatively unreactive, helium stands apart due to its unique duet configuration. Neon, argon, and the other noble gases achieve stability with an octet (eight valence electrons) in their outermost shells. This difference in achieving stability contributes to some subtle differences in their physical and chemical properties, such as boiling points and ionization energies. However, all share the common trait of low reactivity.
The Future of Helium Research
Despite the extensive understanding of helium's properties, research continues to explore its potential applications and delve deeper into its quantum mechanical behavior. With ongoing advancements in cryogenics, superconductivity, and nanotechnology, the demand for helium is likely to increase, highlighting the importance of sustainable helium management and exploration of alternative technologies.
Conclusion: Helium's Unique Valence Electron Count
Helium's possession of only two valence electrons, forming a stable duet, sets it apart from all other elements. This unique configuration directly dictates its exceptional chemical inertness, low boiling point, and low density. These properties are exploited in a wide array of scientific and technological applications, highlighting the importance of this seemingly simple element in modern science and technology. Further research continues to unlock the full potential of helium, promising future advancements in diverse fields. The complete understanding of helium's valence electrons remains a cornerstone of chemistry, providing a fundamental example of electron configuration and its profound impact on an element's properties and behavior.
Latest Posts
Latest Posts
-
Is Tens Of Thousands An Adjective
Apr 03, 2025
-
What Is The Atomic Number Of Ne
Apr 03, 2025
-
4 Light Years In Earth Years
Apr 03, 2025
-
27 To The Power Of 1 3
Apr 03, 2025
-
What Is The Greatest Common Factor Of 54 And 45
Apr 03, 2025
Related Post
Thank you for visiting our website which covers about Number Of Valence Electrons For Helium . We hope the information provided has been useful to you. Feel free to contact us if you have any questions or need further assistance. See you next time and don't miss to bookmark.
