Is Melting Ice Endothermic Or Exothermic
listenit
Apr 05, 2025 · 5 min read
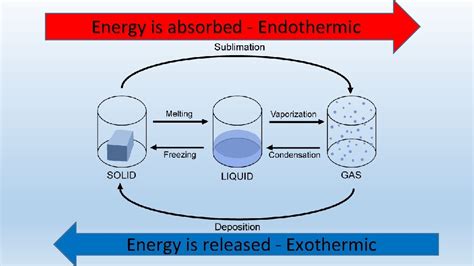
Table of Contents
Is Melting Ice Endothermic or Exothermic? A Deep Dive into Thermodynamics
The question of whether melting ice is endothermic or exothermic is a fundamental concept in thermodynamics, often encountered in chemistry and physics classrooms. While the answer might seem straightforward at first glance, a deeper understanding requires exploring the intricate relationship between energy, phase transitions, and the surrounding environment. This article will delve into the intricacies of this process, providing a comprehensive explanation supported by scientific principles and real-world examples.
Understanding Endothermic and Exothermic Processes
Before we tackle the specifics of melting ice, let's establish a clear understanding of endothermic and exothermic processes. These terms describe the energy exchange between a system (in this case, the ice) and its surroundings.
Endothermic processes absorb heat from their surroundings. This absorption of energy results in a decrease in the temperature of the surroundings. Think of it as the system "drawing in" heat. The energy absorbed is used to overcome the intermolecular forces holding the system together, leading to a change in state or a chemical reaction.
Exothermic processes, conversely, release heat into their surroundings. This release increases the temperature of the surroundings. The system is essentially "giving off" energy. This energy release often occurs as a result of bond formation or a change in state that lowers the overall energy of the system.
The Melting of Ice: An Endothermic Process
Melting ice is unequivocally an endothermic process. To understand why, consider what happens at the molecular level. Ice, in its solid state, consists of water molecules (H₂O) held together by strong hydrogen bonds. These bonds create a relatively ordered crystalline structure.
To transition from a solid (ice) to a liquid (water), these hydrogen bonds must be broken. Breaking these bonds requires energy. This energy is absorbed from the surroundings, hence the endothermic nature of the process. The energy absorbed is used to overcome the attractive forces holding the water molecules rigidly in place within the ice crystal lattice. As energy is absorbed, the molecules gain kinetic energy, increasing their vibrational motion, eventually breaking free from their fixed positions and transitioning into the more disordered liquid phase.
Heat of Fusion: The Energy Required for Melting
The amount of energy required to melt one gram of a substance at its melting point is known as its heat of fusion. For ice, the heat of fusion is approximately 334 joules per gram (J/g). This means it takes 334 joules of energy to melt one gram of ice at 0°C. This energy doesn't increase the temperature of the ice; it solely goes into breaking the hydrogen bonds and facilitating the phase change.
The heat of fusion is a crucial parameter in understanding the endothermic nature of melting. The fact that energy is required to melt ice underscores its endothermic characteristics. If the process were exothermic, it would release heat, which is not the case.
Observational Evidence: Why Melting Ice Cools its Surroundings
The endothermic nature of melting ice is readily observable in everyday life. If you place ice cubes in a drink, the drink gets colder. This is because the ice absorbs heat energy from the drink to melt, causing the drink's temperature to decrease. The ice acts as a heat sink, drawing heat from its surroundings to facilitate the melting process.
Similarly, if you leave a container of ice out in a room, the surrounding air temperature will decrease slightly as the ice melts. This again demonstrates the ice's ability to absorb heat energy from its environment, a clear indication of an endothermic process.
The Role of Temperature and Pressure
While the melting point of ice is typically 0°C at standard atmospheric pressure, changes in pressure can slightly alter this point. However, the fundamental endothermic nature of the process remains unchanged. Increasing the pressure on ice slightly lowers its melting point, but the process still requires energy input to overcome the intermolecular forces and cause the phase transition.
Beyond Ice: Other Endothermic Phase Transitions
The endothermic nature of melting is not unique to ice. The melting of any solid substance is an endothermic process, requiring energy input to overcome the intermolecular or ionic forces holding the solid together. This applies to metals, salts, and a wide range of other materials. The specific heat of fusion varies depending on the substance, reflecting the strength of the intermolecular forces involved.
Practical Applications of Endothermic Melting
The endothermic properties of ice have various practical applications:
- Cooling: The use of ice for cooling beverages and preserving food is a direct application of its endothermic melting properties.
- Refrigeration: Many refrigeration systems utilize the endothermic phase change of refrigerants to cool spaces. These refrigerants absorb heat as they change phase, and this heat is then expelled elsewhere.
- Cryotherapy: In medicine, cold packs containing ice or ice-water mixtures are used in cryotherapy to reduce swelling and inflammation.
Misconceptions About Melting Ice
A common misconception is that the cold from the ice is what causes the cooling effect. While it's true that ice is cold, the cooling effect is primarily due to the endothermic melting process absorbing heat from the surroundings. The ice is already at a low temperature, but it's the energy absorbed during the phase transition that causes the significant cooling effect.
Conclusion: Melting Ice - A Definitive Endothermic Process
In conclusion, melting ice is definitively an endothermic process. It requires an input of energy to overcome the intermolecular forces holding the water molecules in the solid state, enabling the transition to the liquid phase. This energy is absorbed from the surroundings, leading to a decrease in the temperature of the environment. Understanding the endothermic nature of melting ice has significant implications in various fields, ranging from everyday applications to advanced scientific and technological processes. The heat of fusion quantifies the energy requirement, emphasizing the fundamental thermodynamic principle at play. The observable cooling effects in everyday scenarios further solidify the understanding that melting ice is indeed an endothermic process.
Latest Posts
Related Post
Thank you for visiting our website which covers about Is Melting Ice Endothermic Or Exothermic . We hope the information provided has been useful to you. Feel free to contact us if you have any questions or need further assistance. See you next time and don't miss to bookmark.
