Is Magnesium Oxide Ionic Or Covalent
listenit
Apr 02, 2025 · 4 min read
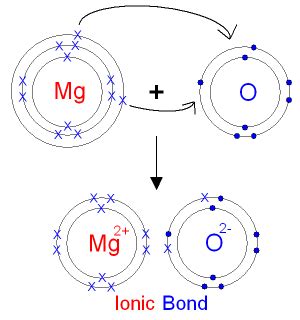
Table of Contents
Is Magnesium Oxide Ionic or Covalent? A Deep Dive into Chemical Bonding
Determining the nature of chemical bonds is fundamental to understanding the properties and behavior of compounds. Magnesium oxide (MgO), a ubiquitous compound with diverse applications, presents a classic example for examining the nuances of ionic versus covalent bonding. This article delves deep into the intricacies of MgO's bonding, exploring its electronegativity difference, crystal structure, and physical properties to definitively answer the question: is magnesium oxide ionic or covalent?
Understanding the Basics of Ionic and Covalent Bonds
Before we dive into the specifics of magnesium oxide, let's briefly review the defining characteristics of ionic and covalent bonds.
Ionic Bonds: The Electrostatic Attraction
Ionic bonds are formed through the electrostatic attraction between oppositely charged ions. This occurs when one atom (typically a metal) donates one or more electrons to another atom (typically a non-metal), creating a positively charged cation and a negatively charged anion. The strong coulombic forces between these ions hold the compound together. Ionic compounds generally exhibit high melting and boiling points, are often brittle, and conduct electricity when dissolved in water or molten.
Covalent Bonds: Shared Electrons
Covalent bonds, on the other hand, involve the sharing of electrons between two atoms. This sharing occurs when atoms have similar electronegativities, meaning they have a comparable attraction for electrons. Covalent compounds typically have lower melting and boiling points than ionic compounds and are often poor conductors of electricity.
Analyzing Magnesium Oxide (MgO): A Case Study
Magnesium oxide (MgO), also known as magnesia, is a white crystalline solid with a high melting point (2852 °C). To determine the nature of its bonding, we need to consider several key factors:
1. Electronegativity Difference: The Driving Force
Electronegativity is a measure of an atom's ability to attract electrons in a chemical bond. The greater the difference in electronegativity between two atoms, the more ionic the bond will be. Magnesium (Mg) has an electronegativity of 1.31, while oxygen (O) has an electronegativity of 3.44. The difference is 2.13, a significant difference. This large electronegativity difference indicates a strong tendency for electron transfer, the hallmark of an ionic bond. Oxygen, being highly electronegative, strongly attracts electrons from magnesium, resulting in the formation of Mg²⁺ cations and O²⁻ anions.
2. Crystal Structure: A Regular Arrangement of Ions
The crystal structure of magnesium oxide provides further evidence for its ionic nature. MgO adopts a rock salt crystal structure, a face-centered cubic arrangement where Mg²⁺ ions and O²⁻ ions are arranged in a regular, alternating pattern. This ordered structure is characteristic of ionic compounds, where the strong electrostatic forces between oppositely charged ions lead to a tightly packed, stable lattice. Covalent compounds, on the other hand, often exhibit more complex and less predictable structures.
3. Physical Properties: High Melting Point and Brittleness
The physical properties of MgO also support the conclusion that it's an ionic compound. Its high melting point (2852 °C) reflects the strong electrostatic attraction between the Mg²⁺ and O²⁻ ions. A significant amount of energy is required to overcome these strong forces and break the ionic bonds. Furthermore, MgO is brittle, a common characteristic of ionic compounds. When stressed, the lattice structure shifts, causing like-charged ions to come into close proximity, resulting in repulsion and fracture.
4. Electrical Conductivity: A Telltale Sign
While solid MgO is a poor conductor of electricity, it becomes a good conductor when molten or dissolved in water. This is because the ions are free to move and carry charge in these states. In the solid state, the ions are fixed in their lattice positions and cannot move freely to conduct electricity. This behavior is typical of ionic compounds.
Addressing Potential Misconceptions
While the evidence strongly suggests that MgO is an ionic compound, some might argue that there's a degree of covalent character involved. This stems from the concept of polarization, where the highly charged cation (Mg²⁺) can distort the electron cloud of the anion (O²⁻). This distortion, though present, is relatively minor in MgO. The large electronegativity difference and the overall dominance of electrostatic forces overshadow any minor covalent character.
Applications of Magnesium Oxide: A Versatile Compound
The unique properties of magnesium oxide make it a versatile compound with a wide range of applications:
- Refractory Materials: Its high melting point makes it ideal for use in high-temperature applications, such as furnace linings and crucibles.
- Medicine: MgO is used as a mild laxative and antacid.
- Agriculture: It's used as a soil amendment to increase its magnesium content.
- Construction: MgO is used in cement and other construction materials.
- Environmental Remediation: MgO can be used to remove pollutants from water and soil.
Conclusion: Magnesium Oxide - Primarily Ionic
Based on the compelling evidence from electronegativity differences, crystal structure, physical properties, and electrical conductivity, we can confidently conclude that magnesium oxide (MgO) is primarily an ionic compound. While a small degree of covalent character may be present due to polarization effects, the dominant interatomic forces are electrostatic attractions between Mg²⁺ and O²⁻ ions. This ionic nature dictates its characteristic properties and its wide range of applications. Understanding the fundamental nature of chemical bonding, as illustrated by MgO, is crucial for comprehending the behavior and application of countless other materials in various fields of science and technology.
Latest Posts
Latest Posts
-
3 Times The Square Root Of 5
Apr 03, 2025
-
What Is 1 Out Of 20 As A Percentage
Apr 03, 2025
-
What Is The Most Common Element Found In Living Things
Apr 03, 2025
-
Horizontal Row In The Periodic Table Are Called
Apr 03, 2025
-
30 Of What Number Is 40
Apr 03, 2025
Related Post
Thank you for visiting our website which covers about Is Magnesium Oxide Ionic Or Covalent . We hope the information provided has been useful to you. Feel free to contact us if you have any questions or need further assistance. See you next time and don't miss to bookmark.
