Ions How Are Ions Made From Neutral Atoms
listenit
Apr 02, 2025 · 6 min read
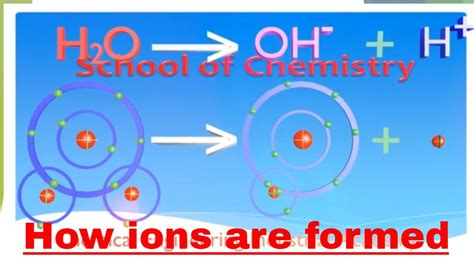
Table of Contents
Ions: How Are Ions Made from Neutral Atoms?
Atoms are the fundamental building blocks of matter, but they don't always exist in a neutral state. Often, atoms gain or lose electrons, transforming into electrically charged particles called ions. Understanding how ions are formed from neutral atoms is crucial to comprehending a vast array of chemical and physical phenomena, from the behavior of electrolytes in our bodies to the workings of advanced technologies like batteries. This article will delve into the process of ion formation, exploring the underlying principles and providing examples to illustrate this fundamental concept in chemistry.
The Structure of an Atom: A Foundation for Ion Formation
Before we dive into ion formation, let's briefly review the structure of a neutral atom. An atom consists of a central nucleus containing positively charged protons and neutral neutrons. Surrounding the nucleus is a cloud of negatively charged electrons, orbiting in specific energy levels or shells. In a neutral atom, the number of protons (atomic number) equals the number of electrons, resulting in a net charge of zero. It's this balance that's disrupted during ion formation.
Electron Shells and Energy Levels
Electrons reside in specific energy levels or shells, each capable of holding a limited number of electrons. The outermost shell, known as the valence shell, plays a crucial role in ion formation. Atoms tend to achieve a stable electron configuration, often resembling that of a noble gas (Group 18 elements), which have a completely filled valence shell. This stability is the driving force behind the gain or loss of electrons, leading to ion formation.
The Formation of Cations: Losing Electrons
Cations are positively charged ions formed when a neutral atom loses one or more electrons. This loss typically occurs when an atom has a relatively small number of electrons in its valence shell, making it easier to lose them than to gain more to fill the shell. The driving force is the attainment of a more stable electron configuration.
Examples of Cation Formation:
-
Sodium (Na): Sodium has one electron in its valence shell. Losing this single electron results in a sodium ion (Na⁺), with a stable electron configuration resembling neon (Ne). This is a common and readily occurring process.
-
Magnesium (Mg): Magnesium has two valence electrons. Losing both electrons forms the magnesium ion (Mg²⁺), achieving a stable electron configuration similar to neon.
-
Aluminum (Al): Aluminum, with three valence electrons, loses all three to form the aluminum ion (Al³⁺), achieving a stable configuration.
The ease with which an atom loses electrons is related to its ionization energy. The ionization energy is the energy required to remove an electron from a neutral atom. Atoms with low ionization energies readily lose electrons and form cations.
The Formation of Anions: Gaining Electrons
Anions are negatively charged ions formed when a neutral atom gains one or more electrons. This happens when an atom has nearly a full valence shell; it's energetically favorable to gain electrons to complete the shell rather than lose many electrons.
Examples of Anion Formation:
-
Chlorine (Cl): Chlorine has seven electrons in its valence shell. Gaining one electron completes its valence shell, forming the chloride ion (Cl⁻), with a stable electron configuration resembling argon (Ar).
-
Oxygen (O): Oxygen has six valence electrons. Gaining two electrons completes its valence shell, forming the oxide ion (O²⁻), also achieving a noble gas configuration.
-
Sulfur (S): Sulfur has six valence electrons, similar to oxygen, and gains two electrons to form the sulfide ion (S²⁻).
The tendency of an atom to gain electrons is related to its electron affinity. Electron affinity is the energy change that occurs when an electron is added to a neutral atom. Atoms with high electron affinities readily gain electrons and form anions.
Factors Influencing Ion Formation
Several factors influence the formation of ions:
-
Electrostatic Forces: The attraction between oppositely charged particles (protons and electrons) is a fundamental force driving ion formation. The loss or gain of electrons alters the balance of these forces, resulting in a net positive or negative charge.
-
Electronegativity: Electronegativity is a measure of an atom's ability to attract electrons in a chemical bond. Atoms with high electronegativity tend to gain electrons and form anions, while those with low electronegativity tend to lose electrons and form cations.
-
Ionic Radius: The size of an ion affects its properties and reactivity. Cations are generally smaller than their parent atoms because the loss of electrons reduces electron-electron repulsion. Anions are generally larger than their parent atoms because the added electrons increase electron-electron repulsion.
-
Chemical Environment: The chemical environment surrounding an atom can influence its tendency to lose or gain electrons. For example, the presence of other charged particles can affect the electrostatic forces involved in ion formation.
The Role of Ions in Chemical Reactions
Ions play a crucial role in various chemical reactions. The electrostatic attraction between cations and anions leads to the formation of ionic compounds, such as sodium chloride (NaCl), commonly known as table salt. These compounds are held together by strong ionic bonds, resulting in crystalline structures.
Ions are also essential components of solutions, where they conduct electricity. These are called electrolytes. Electrolytes are crucial in biological systems, playing vital roles in nerve impulse transmission, muscle contraction, and maintaining fluid balance.
Applications of Ions and Ion Formation
The understanding and application of ion formation have led to significant advancements in various fields:
-
Batteries: Batteries rely on the movement of ions between electrodes to generate electricity. The process involves the oxidation (loss of electrons) at one electrode and reduction (gain of electrons) at the other.
-
Electroplating: Electroplating involves using ions in an electrolytic solution to deposit a thin layer of metal onto a surface. This is used to improve the corrosion resistance, appearance, or other properties of the surface.
-
Corrosion Protection: Understanding ion formation helps in developing strategies to prevent corrosion of metals. Corrosion involves the oxidation of metals, resulting in the formation of metal ions.
-
Medicine: Ions play crucial roles in many physiological processes. Maintaining proper electrolyte balance is essential for human health, and imbalances can lead to serious medical conditions. Ion channels and pumps are crucial in cell biology.
Conclusion: Ions – The Cornerstone of Chemistry and Beyond
Ion formation, a fundamental process in chemistry, involves the gain or loss of electrons by neutral atoms to achieve a more stable electron configuration. This process gives rise to positively charged cations and negatively charged anions, which play pivotal roles in numerous chemical reactions and physical phenomena. The understanding of ion formation is essential for comprehending chemical bonding, electrolyte solutions, battery technologies, and numerous other applications in various fields. The principles governing ion formation, such as ionization energy, electron affinity, and electronegativity, are critical concepts in understanding the behavior of matter at the atomic level and its macroscopic manifestations. From the simplest salt crystal to the complex processes within living cells, ions are ubiquitous and essential for a functioning universe.
Latest Posts
Latest Posts
-
Are Ionic Compounds Solid At Room Temperature
Apr 03, 2025
-
1 Over 5 As A Decimal
Apr 03, 2025
-
60 Percent Of 40 Is What Number
Apr 03, 2025
-
How Does Friction Affect An Objects Motion
Apr 03, 2025
-
How Many Gallons In 12 Pints
Apr 03, 2025
Related Post
Thank you for visiting our website which covers about Ions How Are Ions Made From Neutral Atoms . We hope the information provided has been useful to you. Feel free to contact us if you have any questions or need further assistance. See you next time and don't miss to bookmark.
