Identify The Change Of State Occurring In Each Situation
listenit
Apr 06, 2025 · 7 min read
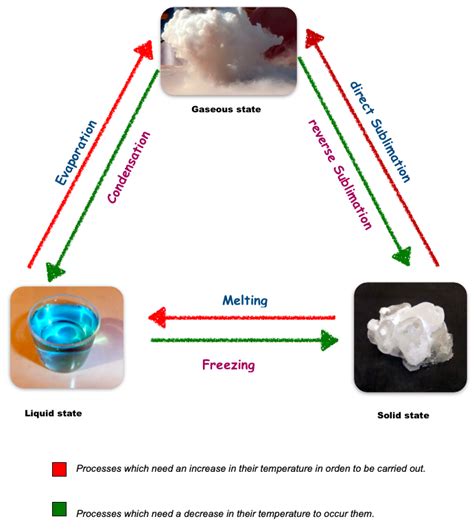
Table of Contents
Identify the Change of State Occurring in Each Situation: A Comprehensive Guide
Changes of state, also known as phase transitions, are fundamental processes in physics and chemistry. They describe the transformation of matter from one state (solid, liquid, gas, or plasma) to another. Understanding these transitions is crucial in various fields, from cooking and meteorology to industrial processes and material science. This article will delve into the different types of changes of state, providing clear explanations and numerous examples to help you identify them in various situations.
The Four Fundamental States of Matter and Their Transitions
Before we explore specific scenarios, let's briefly review the four fundamental states of matter:
-
Solid: Solids have a definite shape and volume. Their particles are tightly packed and have strong intermolecular forces, resulting in a rigid structure.
-
Liquid: Liquids have a definite volume but take the shape of their container. Their particles are close together but can move around more freely than in a solid.
-
Gas: Gases have neither a definite shape nor volume; they expand to fill their container. Their particles are far apart and have weak intermolecular forces.
-
Plasma: Plasma is an electrically charged gas. It's considered the fourth state of matter and is characterized by ionized particles, meaning some electrons have been stripped from their atoms.
Types of Changes of State
There are six main types of changes of state:
1. Melting (Solid to Liquid)
Melting is the process where a solid changes into a liquid due to an increase in temperature. The thermal energy overcomes the intermolecular forces holding the solid's particles together, allowing them to move more freely.
Examples:
- Ice melting into water: A classic example, easily observed on a warm day.
- Chocolate melting in your hand: The warmth of your hand provides the energy needed to melt the chocolate.
- Wax melting in a candle: The heat from the flame causes the wax to transition from a solid to a liquid.
- The process of smelting metals: High temperatures are used to melt metallic ores, allowing for separation and purification.
2. Freezing (Liquid to Solid)
Freezing is the reverse of melting. It's the process where a liquid changes into a solid due to a decrease in temperature. As the temperature drops, the kinetic energy of the particles decreases, allowing intermolecular forces to pull them closer together, forming a rigid structure.
Examples:
- Water freezing into ice: Water solidifies at 0°C (32°F) under standard pressure.
- Molten lava solidifying into rock: The cooling process after a volcanic eruption transforms liquid rock into solid rock formations.
- Making ice cream: The freezing process solidifies the liquid mixture into a delicious frozen dessert.
- Solidification of metal castings: Molten metal is poured into molds and allowed to cool and solidify, forming the desired shape.
3. Vaporization (Liquid to Gas)
Vaporization is the process where a liquid changes into a gas. This can occur in two ways:
- Boiling: Boiling occurs when a liquid is heated to its boiling point, causing rapid vaporization throughout the liquid. Bubbles of vapor form within the liquid and rise to the surface.
- Evaporation: Evaporation occurs at the surface of a liquid at any temperature below the boiling point. The most energetic particles escape the liquid's surface and become gaseous.
Examples:
- Water boiling in a kettle: The heat from the kettle's element causes the water to reach its boiling point and rapidly vaporize.
- Water evaporating from a puddle: On a sunny day, the sun's heat provides the energy for water molecules to escape the puddle's surface.
- Drying clothes: Water evaporates from wet clothes, leaving them dry.
- The production of steam: Water is boiled to create steam, which is used in various industrial processes and power generation.
4. Condensation (Gas to Liquid)
Condensation is the reverse of vaporization. It's the process where a gas changes into a liquid. This typically occurs when a gas cools down, reducing the kinetic energy of its particles and allowing intermolecular forces to draw them closer together.
Examples:
- Dew forming on grass: Water vapor in the air cools down as the temperature drops, condensing into liquid water droplets.
- Fog formation: Water vapor in the air condenses into tiny water droplets, reducing visibility.
- Clouds forming: Water vapor in the atmosphere condenses around microscopic particles, forming clouds.
- Steam condensing on a cold window: The cooler surface of the window causes the water vapor in the air to condense into liquid water.
5. Sublimation (Solid to Gas)
Sublimation is the process where a solid changes directly into a gas without passing through the liquid phase. This occurs when the solid's vapor pressure exceeds the surrounding atmospheric pressure.
Examples:
- Dry ice (solid carbon dioxide) sublimating into carbon dioxide gas: Dry ice doesn't melt; it transitions directly from solid to gas at room temperature.
- Frost disappearing on a cold day: The frost directly changes into water vapor without melting first.
- Freeze-drying of food: This method uses sublimation to remove water from food, preserving its quality.
- Mothballs disappearing over time: The naphthalene in mothballs sublimates into a gas, slowly dissipating the solid.
6. Deposition (Gas to Solid)
Deposition is the reverse of sublimation. It's the process where a gas changes directly into a solid without passing through the liquid phase. This typically occurs when a gas is cooled rapidly below its freezing point.
Examples:
- Frost forming on surfaces: Water vapor in the air directly changes into ice crystals when the temperature drops below freezing.
- Snow formation in clouds: Water vapor in clouds can deposit directly onto ice crystals, contributing to the growth of snowflakes.
- The formation of ice crystals on cold surfaces: On very cold days, water vapor in the air may directly deposit as ice on surfaces.
- Rime ice forming on aircraft: Supercooled water droplets in the air freeze directly onto aircraft surfaces.
Identifying Changes of State in Everyday Situations
Now, let's consider some everyday situations and identify the change of state occurring:
-
A glass of ice water sits on a table on a hot day. The ice melts, and the water evaporates. This involves melting (ice to water) and evaporation (water to water vapor).
-
You put a tray of water in the freezer. The water freezes into ice. This is simply freezing (liquid to solid).
-
Steam rises from a hot cup of coffee. This is vaporization (liquid to gas). Specifically, it's boiling at the surface and evaporation.
-
On a cold morning, you see dew on the grass. This is condensation (gas to liquid).
-
You leave an ice cube out on a countertop, and it shrinks. This is sublimation (solid to gas). Though a small amount may melt first, sublimation is a significant part of the process, as the ice isn't melting into a puddle.
-
Snow accumulates on the ground during a snowstorm. This is deposition (gas to solid).
-
You notice a puddle drying up after a rain shower. This is mostly evaporation (liquid to gas).
-
You see frost on your car windshield in the morning. This is deposition (gas to solid).
-
You bake a cake, and the batter sets into a solid cake. This involves a change of state within the various ingredients.
-
You make chocolate candies, and the melted chocolate solidifies as they cool. This is freezing (liquid to solid).
Factors Affecting Changes of State
Several factors influence the rate at which changes of state occur:
-
Temperature: Higher temperatures generally increase the rate of melting, vaporization, and sublimation, while lower temperatures accelerate freezing, condensation, and deposition.
-
Pressure: Increased pressure generally favors the denser state. For example, higher pressure increases the boiling point of a liquid.
-
Surface area: A larger surface area increases the rate of evaporation and sublimation.
-
Presence of impurities: Impurities can lower the freezing point and raise the boiling point of a substance.
Conclusion
Understanding changes of state is essential for comprehending various natural phenomena and technological processes. By recognizing the different types of transitions and the factors that influence them, you can better interpret the world around you. From the formation of clouds and precipitation to the functioning of refrigerators and industrial processes, changes of state are ubiquitous and profoundly influential. This comprehensive guide aims to provide a solid foundation for identifying and understanding these fundamental changes in the states of matter. Remember to consider the initial and final states and the conditions under which the transition occurs to accurately identify the specific change of state involved.
Latest Posts
Latest Posts
-
What Is 1 66666 As A Fraction
Apr 06, 2025
-
What Is The Si Base Unit For Length
Apr 06, 2025
-
93 Is What Percent Of 186
Apr 06, 2025
-
How To Find All Zeros Of A Polynomial
Apr 06, 2025
-
Domain And Range For X 3
Apr 06, 2025
Related Post
Thank you for visiting our website which covers about Identify The Change Of State Occurring In Each Situation . We hope the information provided has been useful to you. Feel free to contact us if you have any questions or need further assistance. See you next time and don't miss to bookmark.
