How Many Valence Electrons Does O2 Have
listenit
Apr 02, 2025 · 5 min read
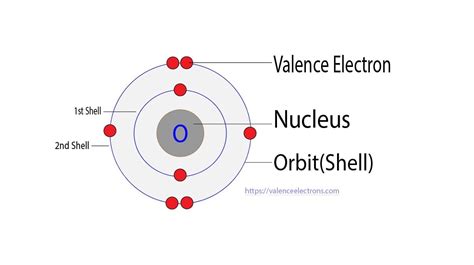
Table of Contents
How Many Valence Electrons Does O2 Have? A Deep Dive into Oxygen's Bonding
Understanding the number of valence electrons in a molecule is crucial for predicting its chemical behavior, bonding properties, and overall reactivity. Oxygen, a vital element for life, exists most commonly as a diatomic molecule, O₂. This article will comprehensively explore the number of valence electrons in O₂, explaining the concept of valence electrons, how to determine them, and the implications for oxygen's chemical behavior. We'll delve into the Lewis structure, molecular orbital theory, and the role of valence electrons in oxygen's reactivity.
Understanding Valence Electrons
Before we dive into the specifics of O₂, let's solidify our understanding of valence electrons. Valence electrons are the electrons located in the outermost shell (also known as the valence shell) of an atom. These electrons are the ones involved in chemical bonding and determine the atom's reactivity. They dictate how an atom will interact with other atoms to form molecules and compounds. The number of valence electrons is directly related to an element's position on the periodic table, specifically its group number (excluding transition metals).
Determining the Valence Electrons of Oxygen (O)
Oxygen (O) is located in Group 16 (or VIA) of the periodic table. This means a neutral oxygen atom possesses six valence electrons. This is because the electronic configuration of oxygen is 1s²2s²2p⁴. The outermost shell (n=2) contains two electrons in the 2s subshell and four electrons in the 2p subshell, totaling six valence electrons.
The O₂ Molecule: A Deeper Look
The oxygen we encounter in the atmosphere exists as a diatomic molecule, O₂. To determine the total number of valence electrons in O₂, we simply add the valence electrons of each oxygen atom. Since each oxygen atom contributes six valence electrons, the O₂ molecule has a total of 12 valence electrons (6 + 6 = 12).
Lewis Structure of O₂: Representing Valence Electrons
The Lewis structure, also known as the electron dot structure, is a simple visual representation of the valence electrons in a molecule. It helps us understand how atoms share electrons to form covalent bonds. While a simple Lewis structure for O₂ might suggest a double bond between the two oxygen atoms, satisfying the octet rule for each oxygen atom, the reality is slightly more complex. This simpler representation accounts for the 12 valence electrons.
.. ..
:Ö=Ö:
.. ..
Beyond the Simple Lewis Structure: Molecular Orbital Theory
The simple Lewis structure, while helpful for a basic understanding, doesn't fully capture the complexity of the O₂ molecule's bonding. Molecular orbital theory (MOT) provides a more accurate description of the bonding in O₂. MOT considers the combination of atomic orbitals to form molecular orbitals, which are occupied by the molecule's electrons.
In O₂, the 2s and 2p atomic orbitals of each oxygen atom combine to form sigma (σ) and pi (π) molecular orbitals. The 12 valence electrons fill these molecular orbitals according to the Aufbau principle and Hund's rule. This results in a double bond between the two oxygen atoms, with two electrons in a σ bonding orbital and four electrons in two degenerate π bonding orbitals. Importantly, there are also two electrons in antibonding π* orbitals, which slightly weaken the bond strength compared to a purely double bond description. This explains the paramagnetism of oxygen; it's slightly attracted to magnetic fields due to the presence of unpaired electrons in the antibonding orbitals.
Implications of Valence Electrons for O₂'s Reactivity
The presence of 12 valence electrons, with unpaired electrons in the antibonding orbitals, significantly influences O₂'s reactivity. Oxygen is a highly reactive molecule, readily participating in oxidation-reduction reactions. Its high electronegativity means it readily accepts electrons to achieve a stable octet configuration. This strong tendency to gain electrons drives its role as an oxidizing agent in numerous chemical processes, including combustion and respiration.
Oxidation and Reduction: The Role of Valence Electrons
The reactivity of O₂ stems directly from its ability to readily accept electrons, using its available valence electron spaces. When O₂ participates in a reaction, it often gains electrons, resulting in a reduction in its overall oxidation state. The substance that donates the electrons to O₂ undergoes oxidation. This fundamental process underlies many biological and industrial reactions.
For example, in the combustion of hydrocarbons (such as methane, CH₄), O₂ acts as an oxidizing agent, accepting electrons from the carbon and hydrogen atoms. This results in the formation of carbon dioxide (CO₂) and water (H₂O), releasing a significant amount of energy. The ability of O₂ to accept electrons, driven by its valence electron configuration, fuels this exothermic reaction.
O₂'s Role in Biological Systems
The crucial role of O₂ in respiration highlights the importance of its valence electrons. In cellular respiration, O₂ acts as the final electron acceptor in the electron transport chain. The electrons, initially derived from the breakdown of glucose, are passed along a series of protein complexes, ultimately reducing O₂ to water. This process generates a substantial amount of ATP, the energy currency of cells, showcasing the crucial role of O₂'s ability to accept electrons in sustaining life.
Conclusion: Valence Electrons and the Reactivity of O₂
In summary, the O₂ molecule possesses 12 valence electrons, a number dictated by the six valence electrons contributed by each oxygen atom. While a simple Lewis structure provides a basic understanding, molecular orbital theory gives a more accurate representation of its electronic structure, including the presence of unpaired electrons in antibonding orbitals. This unique electronic configuration makes O₂ a highly reactive molecule, playing a vital role in numerous chemical reactions, from combustion to respiration. Understanding the number of valence electrons and their distribution in O₂ is essential for understanding its chemical behavior and its critical role in the natural world. The seemingly simple concept of valence electrons is fundamental to explaining the complexity of oxygen's reactivity and its importance in life as we know it.
Latest Posts
Latest Posts
-
How To Find Slope Of Tangent Line At Given Point
Apr 03, 2025
-
How Many Electrons Are In Br
Apr 03, 2025
-
What Is A Kit And Caboodle
Apr 03, 2025
-
Where Is Most Of The Earths Freshwater Found
Apr 03, 2025
-
Write The Sum In Expanded Form
Apr 03, 2025
Related Post
Thank you for visiting our website which covers about How Many Valence Electrons Does O2 Have . We hope the information provided has been useful to you. Feel free to contact us if you have any questions or need further assistance. See you next time and don't miss to bookmark.
