How Many Valence Electrons Are In Mg
listenit
Apr 04, 2025 · 5 min read
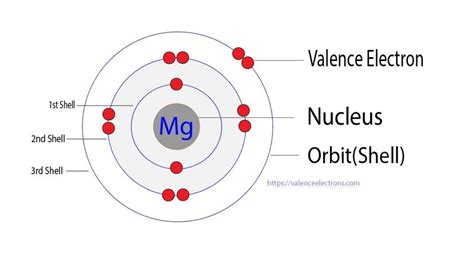
Table of Contents
How Many Valence Electrons are in Mg? A Deep Dive into Magnesium's Electronic Structure
Magnesium (Mg), an alkaline earth metal, plays a crucial role in various biological and industrial processes. Understanding its electronic structure, particularly the number of valence electrons, is fundamental to comprehending its chemical behavior and reactivity. This comprehensive guide will delve into the details of magnesium's valence electrons, exploring its atomic structure, electron configuration, and the implications of its valence electron count for its chemical bonding and properties.
Understanding Valence Electrons
Before diving into the specifics of magnesium, let's establish a clear understanding of what valence electrons are. Valence electrons are the electrons located in the outermost shell of an atom. These electrons are the most loosely bound to the nucleus and, therefore, are the ones primarily involved in chemical bonding. They determine an element's reactivity and the types of chemical bonds it can form. The number of valence electrons dictates an element's position in the periodic table and its chemical properties. Elements in the same group (vertical column) have the same number of valence electrons and, therefore, exhibit similar chemical behavior.
Magnesium's Atomic Structure and Electron Configuration
Magnesium has an atomic number of 12, meaning it possesses 12 protons in its nucleus and, in a neutral atom, 12 electrons orbiting the nucleus. To understand the distribution of these electrons, we need to explore its electron configuration. Electrons occupy energy levels or shells, with each shell having a specific capacity for electrons. The filling of electron shells follows specific rules, primarily the Aufbau principle and Hund's rule.
The electron configuration of magnesium is 1s²2s²2p⁶3s². Let's break this down:
- 1s²: The first shell (n=1) contains the 1s subshell, which can hold up to two electrons. Magnesium has two electrons in this subshell.
- 2s²2p⁶: The second shell (n=2) contains the 2s and 2p subshells. The 2s subshell holds two electrons, and the 2p subshell, which consists of three orbitals, holds six electrons (two electrons per orbital). In total, the second shell has eight electrons.
- 3s²: The third shell (n=3) contains the 3s subshell, which holds the remaining two electrons of magnesium.
Determining the Number of Valence Electrons in Magnesium
From the electron configuration (1s²2s²2p⁶3s²), we can readily identify the valence electrons. The valence electrons are those in the outermost shell, which, for magnesium, is the third shell (n=3). This outermost shell contains two electrons in the 3s subshell.
Therefore, magnesium (Mg) has 2 valence electrons.
Implications of Magnesium's Two Valence Electrons
The presence of two valence electrons significantly influences magnesium's chemical behavior and properties:
-
Reactivity: Magnesium readily loses its two valence electrons to achieve a stable octet configuration (a full outer shell), similar to the noble gas neon. This tendency makes magnesium highly reactive, particularly with nonmetals and other elements that can accept electrons.
-
Oxidation State: Because magnesium loses two electrons, its most common oxidation state is +2. This means that in chemical compounds, magnesium usually exists as a Mg²⁺ ion.
-
Bonding: Magnesium primarily forms ionic bonds, where it transfers its two valence electrons to a more electronegative element, forming an ionic compound. For example, in magnesium oxide (MgO), magnesium donates two electrons to oxygen, forming Mg²⁺ and O²⁻ ions, which are held together by electrostatic attraction.
-
Metallic Bonding: Magnesium also exhibits metallic bonding in its elemental form. The valence electrons are delocalized, forming a "sea" of electrons that surrounds the positively charged magnesium ions. This allows for the characteristic properties of metals, such as electrical and thermal conductivity, malleability, and ductility.
Magnesium's Role in Biological Systems
Magnesium's two valence electrons and resulting chemical properties are crucial for its role in biological systems. Magnesium ions (Mg²⁺) are essential cofactors for numerous enzymes involved in vital metabolic processes, including:
-
DNA replication and repair: Magnesium plays a critical role in the stability of DNA and the processes involved in DNA replication and repair.
-
Protein synthesis: Magnesium is required for the synthesis of proteins, which are essential for various cellular functions.
-
Energy production: Magnesium is involved in the production of ATP (adenosine triphosphate), the primary energy currency of cells.
-
Muscle and nerve function: Magnesium ions are essential for proper muscle contraction and nerve impulse transmission.
Magnesium's Industrial Applications
The unique properties arising from magnesium's two valence electrons make it valuable in various industrial applications:
-
Alloying agent: Magnesium alloys are lightweight and strong, making them ideal for automotive, aerospace, and other applications where weight reduction is crucial.
-
Reducing agent: Magnesium's reducing power is utilized in the extraction of certain metals from their ores.
-
Sacrificial anode: Magnesium's reactivity is exploited in cathodic protection, where magnesium acts as a sacrificial anode to prevent corrosion of other metals.
-
Photographic flash bulbs: Magnesium's ability to burn brightly is used in photographic flash bulbs.
Beyond the Basics: Exploring Advanced Concepts
While the number of valence electrons is a fundamental concept, understanding magnesium's reactivity requires exploring further aspects:
Ionization Energy
Ionization energy is the energy required to remove an electron from an atom or ion. Magnesium's relatively low first and second ionization energies reflect its tendency to readily lose its two valence electrons. However, the subsequent ionization energies are significantly higher, demonstrating the increased difficulty in removing electrons from the inner shells.
Electronegativity
Electronegativity refers to an atom's ability to attract electrons in a chemical bond. Magnesium has a relatively low electronegativity, indicating its tendency to lose electrons rather than gain them. This aligns with its preference for ionic bonding.
Atomic Radius
Magnesium's atomic radius is influenced by the number of electron shells and the effective nuclear charge. Understanding its atomic size is crucial in predicting its interactions with other atoms and molecules.
Conclusion
Magnesium, with its two valence electrons, exhibits characteristic properties and plays a crucial role in both biological and industrial settings. Its reactivity, bonding preferences, and participation in various processes are directly linked to its electronic structure. By understanding the number of valence electrons and their implications, we gain a deeper appreciation for the significance of magnesium in the world around us. The information provided in this article should serve as a comprehensive guide for anyone seeking a thorough understanding of magnesium's electronic structure and its consequences. Further exploration of related concepts such as ionization energy and electronegativity will only enhance this understanding.
Latest Posts
Latest Posts
-
What Is The Highest Common Factor Of 12 And 4
Apr 04, 2025
-
How Many Protons Neutrons Electrons Does Sulfur Have
Apr 04, 2025
-
Is Air A Element Or Compound
Apr 04, 2025
-
What Is 40 Percent Of 5000
Apr 04, 2025
-
How Many Yards Is 180 Inches
Apr 04, 2025
Related Post
Thank you for visiting our website which covers about How Many Valence Electrons Are In Mg . We hope the information provided has been useful to you. Feel free to contact us if you have any questions or need further assistance. See you next time and don't miss to bookmark.
