How Many Protons Neutrons And Electrons Does Nitrogen Have
listenit
Apr 04, 2025 · 5 min read
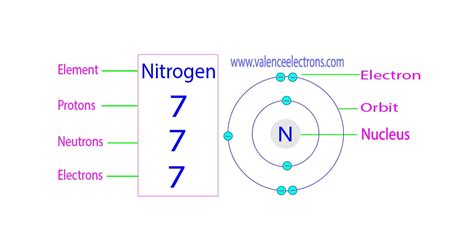
Table of Contents
How Many Protons, Neutrons, and Electrons Does Nitrogen Have? A Deep Dive into Atomic Structure
Nitrogen, a vital element for life as we know it, holds a fascinating place in the periodic table. Understanding its atomic structure—specifically, the number of protons, neutrons, and electrons—is key to grasping its chemical properties and biological significance. This comprehensive guide delves deep into the atomic makeup of nitrogen, exploring related concepts and addressing common misconceptions.
Understanding Atomic Structure: The Building Blocks of Matter
Before we dive into nitrogen's specifics, let's establish a foundational understanding of atomic structure. Atoms, the fundamental units of matter, are composed of three subatomic particles:
- Protons: Positively charged particles located in the atom's nucleus. The number of protons defines the element; it's the atomic number.
- Neutrons: Neutrally charged particles also residing in the nucleus. Neutrons contribute to the atom's mass but not its charge.
- Electrons: Negatively charged particles orbiting the nucleus in electron shells or energy levels. The number of electrons typically equals the number of protons in a neutral atom.
Nitrogen's Atomic Number and Mass Number
Nitrogen's atomic number is 7, meaning it has 7 protons in its nucleus. This is a fundamental characteristic that distinguishes nitrogen from all other elements. This number is unchanging; it's what makes it nitrogen.
The mass number, however, can vary. The mass number is the total number of protons and neutrons in the atom's nucleus. While the number of protons is fixed, the number of neutrons can differ, leading to isotopes.
Isotopes: Variations on a Theme
Isotopes are atoms of the same element (same number of protons) but with different numbers of neutrons. This means they have the same atomic number but different mass numbers. Nitrogen has two stable isotopes found naturally:
- Nitrogen-14 (¹⁴N): This is the most abundant isotope, comprising about 99.6% of naturally occurring nitrogen. It has 7 protons and 7 neutrons (14 - 7 = 7).
- Nitrogen-15 (¹⁵N): This isotope makes up the remaining 0.4% of naturally occurring nitrogen. It has 7 protons and 8 neutrons (15 - 7 = 8).
While other, less stable (radioactive) isotopes of nitrogen exist, they are not naturally abundant and have short half-lives. For most purposes related to chemistry and biology, considering only ¹⁴N and ¹⁵N is sufficient.
Electrons in Nitrogen: Orbitals and Shells
A neutral nitrogen atom has 7 electrons, mirroring its 7 protons. These electrons are distributed in specific energy levels or shells around the nucleus. The electron configuration follows the rules of quantum mechanics, filling orbitals according to the Aufbau principle and Hund's rule:
- First shell (n=1): Contains two electrons in the 1s orbital.
- Second shell (n=2): Contains five electrons. Two are in the 2s orbital, and three are in the 2p orbitals (one electron each in the 2px, 2py, and 2pz orbitals).
This electron configuration is crucial for nitrogen's chemical behavior. The three unpaired electrons in the 2p orbitals make nitrogen highly reactive, readily forming three covalent bonds with other atoms. This is why nitrogen is a crucial component of amino acids, proteins, and nucleic acids – the building blocks of life.
Nitrogen's Role in Biological Systems: A Life-Sustaining Element
Nitrogen's unique atomic structure directly contributes to its vital role in biological systems. The ability to form three covalent bonds allows nitrogen to participate in a vast array of crucial biological molecules:
- Amino acids: The building blocks of proteins, amino acids invariably contain nitrogen atoms in their structures. The amino group (-NH₂) is a hallmark of these essential molecules.
- Nucleic acids (DNA and RNA): Nitrogen is a key component of the nitrogenous bases (adenine, guanine, cytosine, thymine, and uracil) that form the genetic code.
- Chlorophyll: This vital pigment in plants responsible for photosynthesis contains nitrogen atoms within its complex structure.
- Other biomolecules: Nitrogen is found in a wide variety of other biologically important molecules, including hormones, neurotransmitters, and vitamins.
Applications of Nitrogen: Beyond Biology
Beyond its biological significance, nitrogen finds numerous applications in various industries:
- Fertilizers: Ammonia (NH₃), produced from atmospheric nitrogen through the Haber-Bosch process, is a vital component of fertilizers, providing essential nitrogen for plant growth.
- Food preservation: Liquid nitrogen's extremely low temperature is used to freeze food quickly and preserve its quality.
- Electronics: Nitrogen's inert nature makes it useful in the electronics industry to prevent oxidation and corrosion.
- Welding and metallurgy: Nitrogen is used in certain welding processes and in the production of specialized alloys.
Common Misconceptions about Nitrogen's Atomic Structure
Several misconceptions frequently arise concerning nitrogen's atomic structure:
- Confusing atomic number with mass number: It's crucial to remember that the atomic number (number of protons) defines the element, while the mass number (protons + neutrons) can vary within isotopes.
- Ignoring isotopes: Understanding that nitrogen exists in different isotopic forms (¹⁴N and ¹⁵N) is crucial for accurate calculations and applications in fields like isotopic labeling in biological research.
- Misinterpreting electron configuration: A correct understanding of electron shell filling and orbital occupancy is essential for explaining nitrogen's reactivity and bonding behavior.
Conclusion: A Deep Understanding of Nitrogen's Atomic Structure
This comprehensive exploration of nitrogen's atomic structure—its protons, neutrons, and electrons—highlights its fundamental importance in various scientific fields. From its crucial role in biological molecules to its numerous industrial applications, nitrogen's unique atomic properties make it an element worthy of in-depth study. Remembering the key distinctions between atomic number, mass number, and isotopic variations ensures a robust understanding of this indispensable element. The knowledge gained provides a solid foundation for further exploration into the fascinating world of chemistry and its impact on our lives.
Latest Posts
Latest Posts
-
How Many Minute In A Week
Apr 04, 2025
-
How Do Humans Impact Phosphorus Cycle
Apr 04, 2025
-
What The Square Root Of 30
Apr 04, 2025
-
Monomer For Nucleic Acids Is Called
Apr 04, 2025
-
16 And 2 3 As A Fraction
Apr 04, 2025
Related Post
Thank you for visiting our website which covers about How Many Protons Neutrons And Electrons Does Nitrogen Have . We hope the information provided has been useful to you. Feel free to contact us if you have any questions or need further assistance. See you next time and don't miss to bookmark.
