How Many Protons Are In Silver
listenit
Apr 05, 2025 · 5 min read
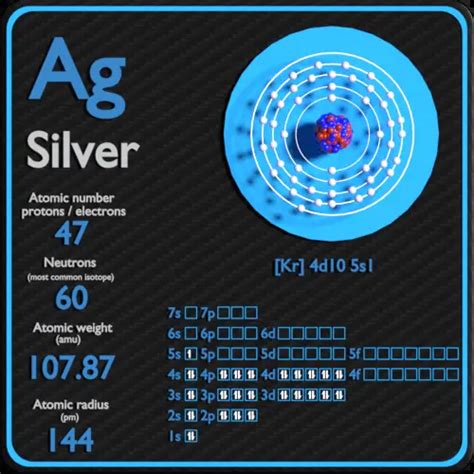
Table of Contents
How Many Protons are in Silver? Understanding Atomic Structure and Silver's Properties
Silver, a lustrous white metal prized for its conductivity and beauty, holds a fascinating place in the periodic table. Understanding its atomic structure, particularly the number of protons it possesses, is key to grasping its unique properties and applications. This article delves deep into the world of silver, exploring its atomic composition, its place in the periodic table, and the significance of its proton count.
Delving into the Atomic Structure of Silver
The foundation of understanding any element lies in its atomic structure. Atoms, the fundamental building blocks of matter, are composed of three subatomic particles: protons, neutrons, and electrons. Protons, positively charged particles, reside in the atom's nucleus. Neutrons, neutral particles, also inhabit the nucleus. Electrons, negatively charged particles, orbit the nucleus in shells or energy levels.
The number of protons in an atom's nucleus defines its atomic number and uniquely identifies the element. This number is crucial because it determines the element's chemical properties and its place on the periodic table. No two elements share the same atomic number.
Silver's Atomic Number: The Key to its Identity
Silver, represented by the symbol Ag (from the Latin argentum), holds the atomic number 47. This means that every silver atom contains exactly 47 protons in its nucleus. This fundamental characteristic is what sets silver apart from all other elements. It's the defining feature that dictates its behavior in chemical reactions and its physical properties.
The Role of Protons in Determining Silver's Properties
The presence of 47 protons within the silver atom's nucleus isn't just a numerical fact; it dictates several key properties of the element:
1. Chemical Reactivity:
The number of protons influences how an atom interacts with other atoms, forming chemical bonds. Silver's 47 protons, along with its electron configuration, contribute to its relatively low reactivity compared to other metals. This is why silver is often found in its pure, elemental form in nature, and why it doesn't readily corrode or react with air and water like some other metals.
2. Electrical Conductivity:
Silver is renowned for its exceptional electrical conductivity – the ability to conduct electrical current efficiently. This property is directly related to its atomic structure. The electrons in the outermost shell of a silver atom are relatively loosely bound, allowing them to move freely within the metallic structure. This free movement of electrons facilitates the efficient flow of electrical charge, making silver an excellent conductor. The 47 protons hold the electrons in the atom, but not so tightly as to impede this vital characteristic.
3. Thermal Conductivity:
Similar to its electrical conductivity, silver's thermal conductivity (ability to conduct heat) is outstanding. The free-moving electrons, influenced by the arrangement determined by the 47 protons, efficiently transfer thermal energy throughout the material. This is why silver is used in applications requiring efficient heat dissipation.
4. Malleability and Ductility:
Silver is highly malleable (can be hammered into thin sheets) and ductile (can be drawn into wires). These properties are a result of the way the silver atoms are arranged and bonded within the metallic structure. The 47 protons in each atom play a critical, albeit indirect, role in defining this metallic bonding, ultimately resulting in its ease of shaping and deformation.
5. Luster and Reflectivity:
Silver's characteristic lustrous appearance and high reflectivity are linked to its interactions with light. The arrangement of electrons, governed by the nuclear charge (number of protons), allows silver to efficiently reflect light across a wide spectrum, contributing to its shiny, metallic look.
Silver's Place in the Periodic Table and its Isotopes
Silver's position in the periodic table (Group 11, Period 5) further reinforces its properties. Its location reflects its electron configuration and chemical behavior. Understanding this placement helps clarify why it behaves the way it does.
Isotopes: Variations in Neutron Count
While the number of protons remains constant (47) in all silver atoms, the number of neutrons can vary. These variations are known as isotopes. The most common isotopes of silver are silver-107 (¹⁰⁷Ag) and silver-109 (¹⁰⁹Ag). Both have 47 protons, but they differ in their neutron count (60 and 62, respectively). This difference in neutron count doesn't significantly affect their chemical properties, but it does affect their atomic mass and some physical properties like density.
Silver's Applications: Leveraging its Unique Properties
The unique combination of properties stemming from its 47 protons makes silver invaluable in various applications:
- Electrical and Electronics: Its excellent electrical conductivity makes it essential in electronics, including circuitry, connectors, and electrical contacts.
- Photography: Silver halides have been historically crucial in photographic film and paper.
- Jewelry and Ornamentation: Its beauty, malleability, and resistance to tarnishing (though not completely resistant) make it a prized material for jewelry and decorative items.
- Medicine: Silver has antibacterial and antimicrobial properties, leading to its use in wound dressings and medical devices.
- Catalysis: Silver's catalytic properties are exploited in certain chemical reactions.
- Mirrors and Coatings: Its high reflectivity makes it ideal for mirrors and reflective coatings.
- Solar Cells: Silver's conductivity plays a role in certain types of solar cells.
Conclusion: The Significance of 47 Protons
The seemingly simple number 47 – the number of protons in a silver atom – holds immense significance. It's the defining characteristic that determines silver's unique properties, shaping its chemical behavior, physical appearance, and diverse applications. Understanding the atomic structure of silver, with its 47 protons at the heart of the atom, allows us to appreciate its importance in our daily lives and numerous technological advancements. From the intricate circuitry of electronics to the lustrous sheen of jewelry, the influence of those 47 protons is undeniable. This deep dive into silver's atomic composition emphasizes the fundamental connection between atomic structure and macroscopic properties, highlighting the beauty and power of scientific inquiry. The seemingly simple fact of 47 protons within a silver atom truly unlocks a world of fascinating properties and applications.
Latest Posts
Latest Posts
-
What Percent Of 752 Is 25
Apr 05, 2025
-
Difference Between Strong Base And Weak Base
Apr 05, 2025
-
What Does An Exclamation Mark Mean In Math
Apr 05, 2025
-
How Many Suns In The Universe
Apr 05, 2025
-
How To Calculate The Net Electric Field
Apr 05, 2025
Related Post
Thank you for visiting our website which covers about How Many Protons Are In Silver . We hope the information provided has been useful to you. Feel free to contact us if you have any questions or need further assistance. See you next time and don't miss to bookmark.
