How Many Molecules Are In 2.50 Moles Of H2o
listenit
Apr 02, 2025 · 5 min read
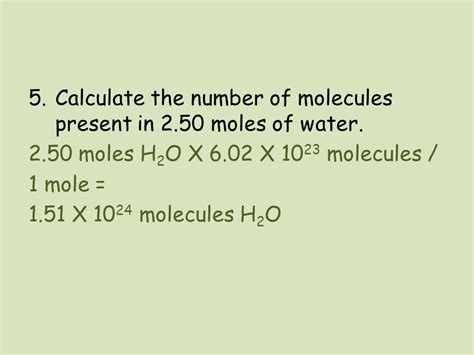
Table of Contents
How Many Molecules Are in 2.50 Moles of H₂O? A Deep Dive into Moles and Avogadro's Number
Understanding the relationship between moles, molecules, and Avogadro's number is fundamental to chemistry. This article will delve into the calculation of the number of molecules in 2.50 moles of water (H₂O), explaining the concepts involved and providing a comprehensive understanding of the process. We'll also explore the broader implications of Avogadro's number and its crucial role in stoichiometry and chemical calculations.
Understanding the Mole Concept
The mole (mol) is a fundamental unit in chemistry, representing a specific amount of substance. It's similar to using a dozen to represent 12 items; a mole represents a specific, enormously large number of particles. This number is known as Avogadro's number, approximately 6.022 x 10²³. One mole of any substance contains Avogadro's number of entities – whether those entities are atoms, molecules, ions, or formula units.
Why Use Moles?
Atoms and molecules are incredibly tiny. Working with individual atoms or molecules is impractical. The mole provides a convenient way to handle large quantities of these microscopic entities, allowing us to relate them to macroscopic measurements like mass and volume. This is essential for performing calculations in chemical reactions and understanding stoichiometry.
Avogadro's Number: The Bridge Between the Macro and Micro Worlds
Avogadro's number is a constant that links the macroscopic world (grams, liters) to the microscopic world (atoms, molecules). It's named after Amedeo Avogadro, an Italian scientist whose work was crucial in developing our understanding of gases and the relationship between volume and the number of particles.
Determining Avogadro's Number
The precise determination of Avogadro's number involves complex experiments, often utilizing X-ray crystallography to determine the precise arrangement of atoms in a crystal lattice. By carefully measuring the mass and volume of a known crystal, scientists can deduce the number of atoms present and, subsequently, calculate Avogadro's number.
Calculating Molecules in 2.50 Moles of H₂O
Now, let's address the central question: how many molecules are in 2.50 moles of H₂O?
The calculation is straightforward:
Number of molecules = (Number of moles) x (Avogadro's number)
-
Identify the given: We are given 2.50 moles of H₂O.
-
Use Avogadro's number: We know that 1 mole contains approximately 6.022 x 10²³ molecules.
-
Perform the calculation:
Number of molecules = 2.50 moles x 6.022 x 10²³ molecules/mole
Number of molecules = 1.5055 x 10²⁴ molecules
Therefore, there are approximately 1.5055 x 10²⁴ molecules in 2.50 moles of H₂O.
Beyond the Calculation: Understanding the Implications
This calculation is more than just a simple arithmetic problem; it highlights the power of the mole concept and Avogadro's number. It allows us to:
- Relate moles to the number of particles: This connection is vital for stoichiometric calculations, enabling us to predict the amounts of reactants and products involved in a chemical reaction.
- Understand macroscopic properties: By knowing the number of molecules, we can begin to understand the macroscopic properties of a substance, linking microscopic behavior to observable characteristics.
- Perform conversions: The mole acts as a central conversion factor, allowing us to convert between moles, mass (using molar mass), volume (using molar volume), and the number of particles.
Molar Mass and its Role in Calculations
The molar mass of a substance is the mass of one mole of that substance, expressed in grams per mole (g/mol). It is crucial for converting between mass and moles. For water (H₂O):
- The atomic mass of hydrogen (H) is approximately 1.01 g/mol.
- The atomic mass of oxygen (O) is approximately 16.00 g/mol.
Therefore, the molar mass of H₂O is: (2 x 1.01 g/mol) + (1 x 16.00 g/mol) = 18.02 g/mol
This means that one mole of water weighs approximately 18.02 grams. We can use this information to calculate the mass of 2.50 moles of water:
Mass = (Number of moles) x (Molar mass) = 2.50 moles x 18.02 g/mol = 45.05 grams
Applications of Avogadro's Number and the Mole Concept
Avogadro's number and the mole concept are fundamental to numerous areas of chemistry and related fields, including:
- Stoichiometry: Calculating reactant and product quantities in chemical reactions.
- Solution chemistry: Determining concentrations of solutions (molarity, molality).
- Gas laws: Relating the volume of a gas to the number of moles present (Ideal Gas Law).
- Thermochemistry: Calculating heat changes in chemical reactions (enthalpy, entropy).
- Analytical chemistry: Quantifying substances in samples using titration and other analytical techniques.
Advanced Considerations: Isotopes and Isotopic Abundance
The calculation we performed assumes that all water molecules are identical. However, this isn't entirely accurate. Hydrogen has two main isotopes: ¹H (protium) and ²H (deuterium). Oxygen also has several isotopes, the most common being ¹⁶O, ¹⁷O, and ¹⁸O. The presence of these isotopes slightly alters the average molar mass of water, leading to minor variations in the actual number of molecules present in a given mass or number of moles. However, for most practical purposes, the approximation using the average molar mass is sufficiently accurate.
Conclusion: The Power of the Mole
The mole concept and Avogadro's number are indispensable tools in chemistry. They bridge the gap between the microscopic world of atoms and molecules and the macroscopic world of observable quantities. Understanding these concepts is crucial for mastering stoichiometry, solution chemistry, and numerous other areas of chemical study. The seemingly simple calculation of the number of molecules in 2.50 moles of H₂O exemplifies the power and importance of this fundamental concept in our understanding of the chemical world. Through a clear understanding of these principles, we gain invaluable insight into the quantitative aspects of chemical reactions and the behavior of matter. This foundation allows for more advanced exploration into the fascinating intricacies of chemistry and its far-reaching applications.
Latest Posts
Latest Posts
-
What Percent Is 3 Out Of 20
Apr 03, 2025
-
500 Ml Of Water Is How Many Ounces
Apr 03, 2025
-
What Is 9 Ounces In Cups
Apr 03, 2025
-
Explain Why Chemical Equations Have To Be Balanced
Apr 03, 2025
-
How Do You Write 10 As A Fraction
Apr 03, 2025
Related Post
Thank you for visiting our website which covers about How Many Molecules Are In 2.50 Moles Of H2o . We hope the information provided has been useful to you. Feel free to contact us if you have any questions or need further assistance. See you next time and don't miss to bookmark.
