How Many Electrons Can Be Held In The Third Orbital
listenit
Apr 05, 2025 · 5 min read
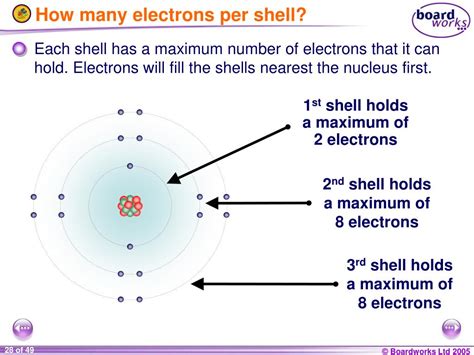
Table of Contents
How Many Electrons Can Be Held in the Third Orbital? A Deep Dive into Electron Configuration
Understanding electron configuration is fundamental to grasping the behavior of atoms and molecules. A key aspect of this is knowing how many electrons each orbital can hold. This article delves into the specifics of the third orbital, explaining not just the number of electrons it can accommodate, but also the underlying principles governing electron arrangement within an atom.
Understanding Electron Shells, Subshells, and Orbitals
Before we dive into the third orbital, let's clarify the hierarchical structure of electron arrangement within an atom.
Electron Shells: Energy Levels
Electrons reside in various energy levels, also known as electron shells or principal energy levels. These shells are designated by the principal quantum number, n, where n = 1, 2, 3, and so on. The lower the value of n, the closer the electrons are to the nucleus and the lower their energy. The first shell (n = 1) is closest to the nucleus, followed by the second shell (n = 2), the third shell (n = 3), and so forth.
Electron Subshells: Defining Orbital Shapes
Each electron shell is further divided into subshells, which are regions of space with slightly different energies within the same shell. These subshells are characterized by the azimuthal quantum number, l, which can take on integer values from 0 to n - 1. Each value of l corresponds to a specific subshell:
- l = 0: s subshell (spherical shape)
- l = 1: p subshell (dumbbell shape)
- l = 2: d subshell (more complex, multi-lobed shape)
- l = 3: f subshell (even more complex shape)
Electron Orbitals: Specific Regions of Probability
Within each subshell, there are one or more orbitals. An orbital is a region of space where there's a high probability of finding an electron. The number of orbitals within a subshell is determined by the magnetic quantum number, ml, which can take on integer values from -l to +l, including 0. This means:
- s subshell: 1 orbital (ml = 0)
- p subshell: 3 orbitals (ml = -1, 0, +1)
- d subshell: 5 orbitals (ml = -2, -1, 0, +1, +2)
- f subshell: 7 orbitals (ml = -3, -2, -1, 0, +1, +2, +3)
The Third Electron Shell (n=3)
Now, let's focus on the third electron shell (n = 3). According to the rules outlined above, the third shell contains three subshells:
- 3s subshell: One 3s orbital (l=0, ml=0)
- 3p subshell: Three 3p orbitals (l=1, ml = -1, 0, +1)
- 3d subshell: Five 3d orbitals (l=2, ml = -2, -1, 0, +1, +2)
How Many Electrons Can the Third Shell Hold?
Each orbital can hold a maximum of two electrons, according to the Pauli Exclusion Principle. This principle states that no two electrons in an atom can have the same set of four quantum numbers (n, l, ml, and ms, where ms is the spin quantum number, representing the electron's spin, either +1/2 or -1/2).
Therefore, the total number of electrons the third shell can hold is calculated as follows:
- 3s subshell: 1 orbital * 2 electrons/orbital = 2 electrons
- 3p subshell: 3 orbitals * 2 electrons/orbital = 6 electrons
- 3d subshell: 5 orbitals * 2 electrons/orbital = 10 electrons
Total electrons in the third shell: 2 + 6 + 10 = 18 electrons
Consequently, the maximum number of electrons that can occupy the third electron shell is 18.
Filling Orbitals: The Aufbau Principle and Hund's Rule
The order in which electrons fill orbitals isn't completely arbitrary. Two fundamental principles guide this process:
The Aufbau Principle
The Aufbau principle states that electrons fill atomic orbitals of the lowest available energy levels before occupying higher levels. In general, the order of filling is:
1s, 2s, 2p, 3s, 3p, 4s, 3d, 4p, 5s, 4d, 5p, 6s, 4f, 5d, 6p, 7s, 5f, 6d, 7p...
Note the slight deviation from a strictly increasing n value. This is due to subtle energy level variations between subshells.
Hund's Rule
Hund's rule states that electrons will individually occupy each orbital within a subshell before doubling up in any one orbital. This minimizes electron-electron repulsion and leads to a more stable configuration. Electrons in singly occupied orbitals will have parallel spins (+1/2).
Examples of Electron Configurations in the Third Shell
Let's examine the electron configurations of a few elements to illustrate how the third shell is populated:
-
Sodium (Na, atomic number 11): 1s²2s²2p⁶3s¹ Sodium has one electron in the 3s orbital of its third shell.
-
Chlorine (Cl, atomic number 17): 1s²2s²2p⁶3s²3p⁵ Chlorine fills the 3s and partially fills the 3p subshells in its third shell.
-
Argon (Ar, atomic number 18): 1s²2s²2p⁶3s²3p⁶ Argon completely fills both the 3s and 3p subshells in its third shell.
-
Zinc (Zn, atomic number 30): 1s²2s²2p⁶3s²3p⁶4s²3d¹⁰ Zinc completely fills the third shell with its 18 electrons, demonstrating the maximum capacity.
Beyond the Third Shell: Implications for Chemical Behavior
The electron configuration of an atom, especially the arrangement of electrons in its outermost shell (valence electrons), dictates its chemical properties and reactivity. Atoms tend to react in ways that achieve a stable electron configuration, often resembling a noble gas (completely filled outer shell). Understanding the electron capacity of the third shell is crucial to predicting how elements in the third period of the periodic table will interact chemically. For instance, elements with partially filled 3s or 3p subshells tend to be more reactive than those with a complete third shell.
Conclusion
The third electron shell, with its capacity to hold a maximum of 18 electrons, plays a crucial role in determining the properties and reactivity of a wide range of elements. By understanding the principles governing electron configuration, including the Aufbau principle, Hund's rule, and the Pauli exclusion principle, we can accurately predict and explain the behavior of atoms and molecules. This knowledge forms the foundation of many chemical concepts and is essential for further study in chemistry and related fields. This article comprehensively details the third orbital electron capacity and the underlying principles governing electron arrangement in atoms, offering a complete understanding of this essential concept in chemistry.
Latest Posts
Latest Posts
-
Where Is The Most Freshwater On Earth Found
Apr 06, 2025
-
Highest Common Factor Of 20 And 36
Apr 06, 2025
-
How Many Valence Electrons Are In Ne
Apr 06, 2025
-
What Is Meant By Change Of State
Apr 06, 2025
-
How Many Orbitals Does The D Subshell Have
Apr 06, 2025
Related Post
Thank you for visiting our website which covers about How Many Electrons Can Be Held In The Third Orbital . We hope the information provided has been useful to you. Feel free to contact us if you have any questions or need further assistance. See you next time and don't miss to bookmark.
